Lunar and Martian Regolith Simulants Desorb and Weather after Exposure to Bioregenerative Life Support System Effluent
ACS Earth Space Chem. 2026, 10, 2, 441–454: Graphical abstract
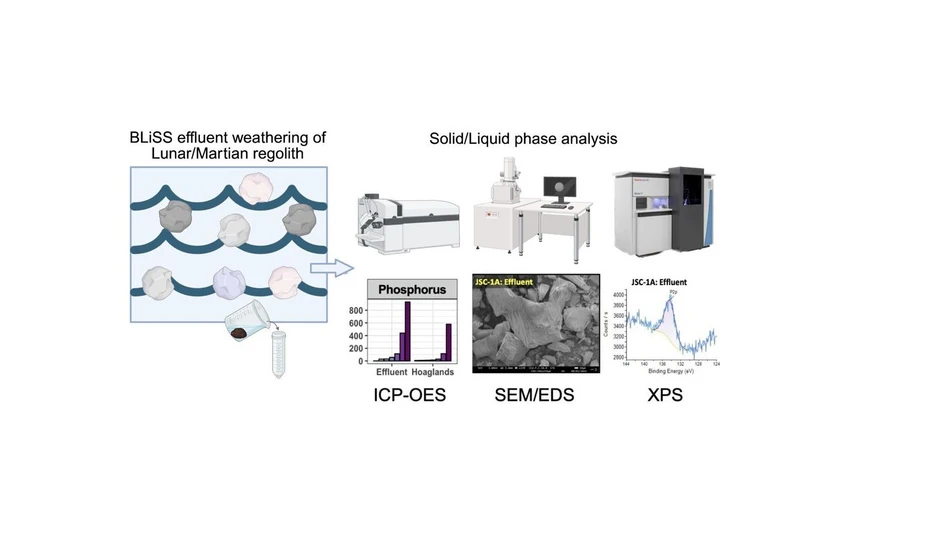
This study investigates nutrient exchange between lunar and martian regolith simulants and bioregenerative life support system (BLiSS) effluent. Using ICP-OES, sorption and dissolution processes were quantified, revealing significant release of elements such as S, Ca, Mg, and Na from the simulants.
Surface and structural analyses confirmed elemental bonding and mineral alterations after reaction. The findings demonstrate that regolith can supply essential nutrients to BLiSS systems, supporting sustainable agriculture in extraterrestrial environments and highlighting the importance of realistic effluent simulations.
The original article
Lunar and Martian Regolith Simulants Desorb and Weather after Exposure to Bioregenerative Life Support System Effluent
Harrison R. Coker, Daniella Saetta, Misle M. Tessema, Jackson L. Smith, Charles A. Richardson-Gongora, Jason A. Fischer, Hannah I. Roberts, Luke B. Roberson, and Julie A. Howe*
ACS Earth Space Chem. 2026, 10, 2, 441–454
https://doi.org/10.1021/acsearthspacechem.5c00267
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
Background
Inorganic nutrient solutions (e.g., Hoagland’s (1)) are useful for growing plants in soilless systems but require continuous nutrient inputs that are consumed as plants grow. With future plans for lunar and Mars settlements, dependency on Earth-sourced fertilizers will hinder efficient operations. Therefore, extracting nutrients from regolith to enhance sustainable agricultural operations, or improving the regolith’s fertility such that plants directly derive nutrients from the regolith, are both effective strategies that support diverse cropping operations to produce food and fiber resources through in situ resource utilization (ISRU). (2,3) Broadly, approaches that utilize surface resources from the Moon and Mars to reduce inputs from Earth are deemed ISRU. (4)
In Situ Resource Utilization
The extraction of raw elements from regolith is a primary goal of ISRU. (4) Various approaches for enhanced dissolution of regolith have been studied, (5) including techniques such as ionic liquids, (6) eutectic salts, (7) electro-deoxidation, (8) and heat treatment. (9) While promising, these approaches have the downfall of requiring exogenous chemicals, energy, and technology. Alternatively, a basic approach will encompass using resources such as BLiSS effluent to physically weather the regolith. Because of the underlying basaltic mineralogy of lunar and martian regoliths, hydrologic weathering may be a rapid process that deserves further investigation.
Bioregenerative Life Support Systems
To recycle consumed nutrients from undesirable plant and human wastes, the use of bioregenerative life support systems (BLiSS) that decompose organic matter into inorganic nutrient streams will be used. (10) Organic wastes (e.g., plant material, human wastes, etc.) generated by lunar and martian astronauts will necessitate recycling via a series of BLiSS bioreactors that result in effluent streams of water, inorganic nutrients, and metabolites of anerobic digestion, which include various volatile organic acids. (11) A working BLiSS prototype at Kennedy Space Center (KSC), called the organic processing assembly (OPA), utilizes dual-stage anerobic bioreactors and membrane filtration (12,13) to accomplish decomposition of organic matter, which is subsequently fed to a phototrophic membrane bioreactor (14) for the complete oxidation of N species. The microbiome of OPA varies across reactors as a function of varying inputs and substrate decomposition. (15) While OPA is a developing technology in both biological processing and technological command, analogous BLiSS, will be required to create a circular economy of nutrients in space. (16) Because BLiSS produces nutrient streams that differ from those of inorganic nutrient solutions, investigating fundamental interactions of BLiSS effluent with regolith simulants will help guide future planning and determine uses of effluent for outposts on the Moon and Mars. In this study, effluent from the OPA at KSC was utilized, as it is a high-fidelity prototype of what may be produced in spaceflight and on lunar and martian outposts.
Lunar and Martian Regolith
Lunar regolith is relatively homogeneous in elemental composition, with the major difference being the abundance of Ca. A useful classification refers to lunar samples as being “high-Ca” or “low-Ca” above or below 13.5 wt % CaO. (17) Increasing abundance of nanophase Fe, along with decreasing grain size, is characteristic of more mature lunar regolith. (18) Lunar samples exhibit low electrical conductivity (EC), though the electrical properties of lunar regolith are especially sensitive to thermal and atmospheric changes. (19,20) In general, the chemical reactivity of lunar regolith to organic molecules is not well understood, (21) but as aqueous and oxidizing environments are introduced to support plant life, it should be expected that lunar regolith will drastically change. For instance, exposure to water vapor increases the adsorption of N2 and Ar, (22) while the crushing of regolith promotes the movement of trapped vaporous gases into solution. (23) Lunar simulants have additionally demonstrated increased generation of reactive oxygen species (ROS) (e.g., H2O2 and OH–) in micromolar quantities when crushed and exposed to the atmosphere. (24) Lunar regolith contains up to 70% agglutinates by volume, (25) which are aggregates of mineral/lithic fragments fused together by rapid melting and cooling of glass after meteoroid impacts. Agglutinates are noted to have differing abundances of nanophase Fe0, with several hypotheses accounting for their incorporation into the lunar regolith. (26)
Martian astronauts will have greater accessibility to specific and diverse minerals for ISRU than lunar astronauts. For Mars, there is likely a semiactive hydrologic cycle of hydrated brines (27−30) that may even support microbial life. (31) Although glass is a more common feature of lunar regolith, in analog martian sites on Earth, glass-matrices have been identified as the possible profile-controlling minerals. (32) Martian regolith is high in Fe metal oxides, (33) chloride, (34) and sulfate salts, (35) including perchlorates (ClO4–) and other oxidants, such as hydrogen peroxide (H2O2), which occur at toxic levels for plants. These salts may limit bioavailability of various metals. (36) Oxidized nitrogen species, simple organics, oxychlorine phases, and sulfates are widespread, with phyllosilicates and carbonates occurring in select Gale Crater materials, and it appears that geochemical conditions and organic C may have once been favorable for microbial life. (37,38) Recent evidence from the Phoenix landing site quantified organic C at 83–1484 μg C g–1 and carbonates from 1.1 to 2.6 wt %. (39) At Gale Crater, the 2007 Phoenix Mars Scout Mission’s Wet Chemistry Laboratory (WCL) conducted in situ experiments at the Phoenix landing site and found solutions were dominated by ClO4–, Mg2+, and Na+, at mM levels, with sub-mM concentrations of Ca2+, K+, and Cl– and a pH of 7.7.34 The minimal leachable ions from surface samples had a corrected electrical conductivity (EC) ranging from 1370 to 1900 μS cm–1 at 25 °C. As the WCL extraction cell measured soil extracts at 1:25 (solid/solution), the reported EC values would likely be 5–25 times higher using traditional soil approaches (saturated paste extract; 1:1–1:5), indicating a highly saline soil that will need to be remediated prior to being used as a plant growth substrate, of which leaching with BLiSS solutions may be an effective management strategy.
Methods
Analyses
After the 24 h batch experiment, the pH and electrical conductivity (EC) of the reacting solutions were measured to assess changes to solution chemistry after weathering. Solutions were acidified to 1% HNO3 and analyzed via inductively coupled plasma-optical emission spectroscopy (ICP-OES) (Thermo Scientific, iCAP 7000) in duplicate. Ion chromatography was used to analyze the ions without any sample preparation. Substrate samples were Au sputter-coated (Denton Vacuum, Desk IV) and mounted prior to scanning electron microscope-energy dispersive X-ray spectroscopy (SEM-EDS) (Jeol, JSM-IT800). X-ray photoelectron spectroscopy (XPS) (Thermo Scientific, Nexsa G2) was used on substrates to identify elemental bonding associations by using Qtegra software. XPS data were processed in the Avantage Data System. X-ray diffraction (XRD) (Malvern Panalytical, Empyrean) was used to assess mineralogical changes to substrates and was performed using Bragg–Brentano mode. The diffraction data were recorded from 15 to 70 two-theta (2θ) degrees. HighScore curve fitting software was used to process the diffraction pattern and compared to the PDF-4+2023 International Center of Diffraction Database (ICDD).
Results
Weathering of Substrates
Visual observations of the substrates after the batch experiment revealed weathering to the minerals. In JSC-1A, the sharp mineral edges appeared to have some rounded-off features that reduced the overall sharpness of corners and edges (Figure 5); however, a quantitative analysis was not available to indicate if mineral sharpness was statistically reduced. In general, the overall particle size was not substantially reduced in a survey of all of the minerals. Notable artifacts of the weathering process include holes in the mineral faces where small particles collapsed and the appearance of webbing patterns in the anorthosite fraction of JSC-1A. Unfortunately, EDS was not able to detect any sorbed elements in these features (Figure 6). Because the detection limits of EDS are ∼1000 mg kg–1, it is reasonable that no sorbed features were detected, given that measured sorption densities after batch experimentation were lower than EDS detection limits. Further, the probed volume at a given electron energy is below the surface, whereas adsorbents are likely only located on the mineral surface.
 ACS Earth Space Chem. 2026, 10, 2, 441–454: Figure 5. Lunar simulant JSC-1A imaged with scanning electron microscope (SEM). The electron beam energy ranged from 1 to 3 kV. The Hoagland’s and effluent images are after the 24 h batch experiment.
ACS Earth Space Chem. 2026, 10, 2, 441–454: Figure 5. Lunar simulant JSC-1A imaged with scanning electron microscope (SEM). The electron beam energy ranged from 1 to 3 kV. The Hoagland’s and effluent images are after the 24 h batch experiment.
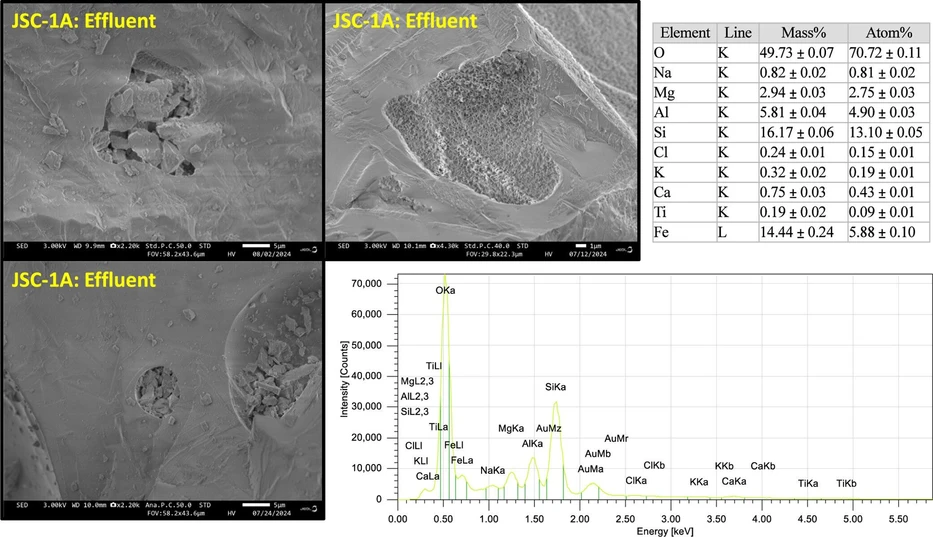 ACS Earth Space Chem. 2026, 10, 2, 441–454: Figure 6. Scanning electron microscope (SEM) image of erosion spots on lunar simulant JSC-1A after batch experiment. Electron dispersive spectroscopy (EDS) was used to investigate the elemental abundance of erosion regions and was set to 30 keV.
ACS Earth Space Chem. 2026, 10, 2, 441–454: Figure 6. Scanning electron microscope (SEM) image of erosion spots on lunar simulant JSC-1A after batch experiment. Electron dispersive spectroscopy (EDS) was used to investigate the elemental abundance of erosion regions and was set to 30 keV.
Chemical Interaction
The elements C, N, P, and Ca were investigated with high-resolution XPS. For C, a major peak at ∼286 eV was observed in all substrates and solutions that corresponded to C–O bonding. Hoagland’s and effluent did not affect the XPS spectra significantly in JSC-1A; however, in MGS-1, there was a notable increase in the Hoagland’s and effluent-treated regolith at ∼294 eV, which corresponds to CF3 (Figure 9). Because F was not quantified, it is difficult to know if F was in differing concentrations between solutions. Hoagland’s was made from DI water and effluent processes a mixture of fecal simulant and tap water; it is unlikely that F was greater in Hoagland’s.
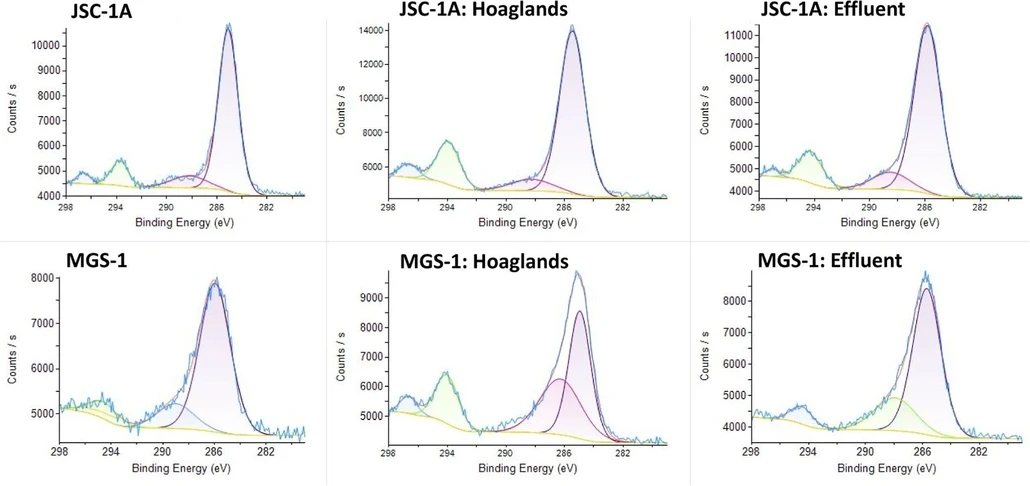 ACS Earth Space Chem. 2026, 10, 2, 441–454: Figure 9. Carbon (C 1s) peaks of lunar (JSC-1A) and martian (MGS-1) simulants as unaltered materials and after batch experiment using XPS.
ACS Earth Space Chem. 2026, 10, 2, 441–454: Figure 9. Carbon (C 1s) peaks of lunar (JSC-1A) and martian (MGS-1) simulants as unaltered materials and after batch experiment using XPS.
Conclusion
The hydrologic weathering of lunar and martian regolith simulants by BLiSS streams demonstrated increased plant essential nutrients and metals in solution due to highly soluble compounds and easily weatherable mineralogy. Multielement competitive sorption demonstrates low removal of P and K, but high supplementation of Ca, Mg, and S to the solution. The mechanistic differences between the organic BLiSS effluent and the inorganic nutrition solution were predominantly unaccounted for. It stands to reason that longer-term studies may follow up with the provided results to fully determine how supplementation of BLiSS solutions can be obtained from weathering of the regolith. Therefore, further experimentation that improves upon the extraction of elements of interest and increases the reaction time will be useful in fortifying the sustainability of human operations. The hydrologic weathering of sharp mineral edges also deserves quantitative investigation over time to understand whether reducing the abrasiveness of regolith offers an additional aspect of using BLiSS effluent on lunar and martian surface minerals.