Single-atom-enhanced membrane for simultaneous bacteria and heavy metal on-site water treatment

CHEMJOURNAL-D-25-00469: Figure 1. Schematic of the one-step water filtration process using Mn-NGCOOH and NGCOOH layers. The illustration shows water flowing through the membrane with simultaneous removal of bacteria and heavy metals. Insets highlight carboxyl-metal binding, manganese coordination on the graphene surface, bacterial membrane structure, and the trapping of bacteria through Mn2+ binding to outer-membrane polysaccharides.
This study presents a lightweight, graphene-based membrane engineered with single atoms for simultaneous removal of bacteria and heavy metals from water without electricity or complex infrastructure.
The membrane achieves >99.999% bacterial removal and high heavy metal adsorption while maintaining performance over multiple reuse cycles, offering a practical solution for off-grid and emergency water purification.
The original article
Single-atom-enhanced membrane for simultaneous bacteria and heavy metal on-site water treatment
David Panáček, Renata Večeřová, Zdeněk Baďura, Vítězslav Hrubý, Lukáš Zdražil, Milan Kolář, Thamraa Alshahrani, Aleš Panáček, Aristides Bakandritsos, Radek Zbořil
CHEMJOURNAL-D-25-00469
https://doi.org/10.1016/j.chempr.2025.102785
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
Exploring new approaches for the effective purification of contaminated water is a key research area.40,41,42 Cutting-edge technologies for bacteria decontamination are related to establishing a strong binding between the filtration material and bacteria (mainly to the organic groups on the outer layer of the bacterial membrane.21,43,44) Among such sophisticated purification methods is a system inspired by the water transport and transpiration of trees.45 The material (composed of CS, hydroxyapatite nanowires, and palladium nanoparticles) functions via the interaction between the negatively charged bacteria and the positively charged CS component, leading to the arrest of their vital functions (e.g., movement and reproduction) and thus to their death. Another approach involves the use of superparamagnetic nickel nanocrystals, rendering the ability to bind to Gram-positive and Gram-negative bacteria.21 However, a potential limitation of such systems is the use of toxic elements such as palladium and nickel. Moreover, achieving an integrated system that combines effective and safe bacterial disinfection with heavy metal decontamination in a single step remains an unmet challenge. Although nanoparticles have enabled major advances in water treatment,10,21,32 they often face inherent limitations arising from heterogeneous surfaces, wide size distributions, and aggregation under operating conditions.46,47 These issues reduce accessible surface area, limit catalytic efficiency, and introduce variability, undermining reproducibility and long-term performance.46 In contrast, single-atom catalysts (SACs) represent a fundamental advance, delivering maximal atomic efficiency, uniform coordination, and precisely localized active sites on suitable supports.48,49 This atomic dispersion prevents aggregation and enables tunable local chemistry, affording precise control over binding affinity, redox behavior, and reaction selectivity.40 These features enable high catalytic performance in decontamination processes, including Fenton-like mechanisms,50 ROS production,51 and selective multivalent coordination to pollutants.44 As a result, SACs combine catalytic efficiency with enhanced sustainability and safety by minimizing toxic metal use and leaching. Their development represents a key opportunity toward precision-designed water purification systems that integrate performance, selectivity, and environmental compatibility.
Inspired by these exceptional properties, we engineered a dual-functionalized graphene derivative loaded with single atoms of manganese and carboxylic groups (Mn-NGCOOH) as a biocompatible and very efficient trap for heavy metals (Cd2+ and Pb2+) and bacteria (Figure 1). Membranes prepared from this material were applied as a filtration system exhibiting exceptional efficiency in removing bacterial and heavy metal contaminants from various water sources, including distilled, tap, and river water. The membrane demonstrated exceptional filtration efficiency (>99.999%) for bacterial removal across various water sources while also exhibiting high adsorption capacity for heavy metals. Furthermore, the filtration system retains over 90% efficiency after six regeneration cycles, ensuring cost-effectiveness and long-term sustainability. The simple operation and high membrane permeability, requiring only a hand-powered vacuum, ensure its applicability in remote areas without any power source, representing a substantial advancement in portable water purification technologies.
 CHEMJOURNAL-D-25-00469: Figure 1. Schematic of the one-step water filtration process using Mn-NGCOOH and NGCOOH layers. The illustration shows water flowing through the membrane with simultaneous removal of bacteria and heavy metals. Insets highlight carboxyl-metal binding, manganese coordination on the graphene surface, bacterial membrane structure, and the trapping of bacteria through Mn2+ binding to outer-membrane polysaccharides.
CHEMJOURNAL-D-25-00469: Figure 1. Schematic of the one-step water filtration process using Mn-NGCOOH and NGCOOH layers. The illustration shows water flowing through the membrane with simultaneous removal of bacteria and heavy metals. Insets highlight carboxyl-metal binding, manganese coordination on the graphene surface, bacterial membrane structure, and the trapping of bacteria through Mn2+ binding to outer-membrane polysaccharides.
Results
Single-atom-enhanced filtration: Synthesis, structure, and filter construction
The filtration device was designed in three key steps: (1) synthesis of nitrogen-doped graphene equipped with carboxyl groups (NGCOOH), (2) coordination of Mn2+ ions with NGCOOH (Mn-NGCOOH), (3) assembly of the filtration membrane by integrating NGCOOH and Mn-NGCOOH (Figure 1 and Methods). Briefly, fluorographene reacted with sodium azide and then with nitric acid to afford the NGCOOH. X-ray photoelectron spectroscopy (XPS) showed extensive defluorination (0.37 atom %) and nitrogen doping (5.52 atom %) upon the transformation of fluorographene to NGCOOH. In addition, NGCOOH exhibits a high oxygen content (23.73%), predominantly in the form of carboxyl groups (Figures S1A and S2A), which are essential for the binding of metal atoms to its surface.25,28 These structural features were also confirmed by Fourier transform infrared spectroscopy (FTIR; Figures 2A and 2B). The FTIR spectra of NGCOOH and Mn-NGCOOH exhibited features similar to common organic carboxylic acids, particularly the broad absorption band of O–H stretching between 2,700 and 3,700 cm−1 due to excessive hydrogen bonding of the groups. The absorption bands at 3,474 and 3,240 cm−1 correspond to various H-bonding configurations.52 The strong band at 1,720 cm−1 is typical of the carboxylic groups.52 Symmetric stretching vibration bands at 1,430 and 1,345 cm−1, as well as C–O stretching at 1,245 cm−1, appear at the same region with a broad feature of skeletal aromatic C=C vibrations.52 Immobilization of Mn2+ on NGCOOH resulted in a loading of 2 wt %, as determined by inductively coupled plasma mass spectrometry (ICP-MS). The carboxyl groups detected by FTIR were partially ionized to carboxylates after interaction with Mn2+, as indicated by the occurrence of vibrations at 1,600 and 1,430/1,345 cm−1 in Mn-NGCOOH (Figures 2A and 2B). Detailed characterization of NGCOOH and Mn-NGCOOH is available in our previous work by Panáček et al.44
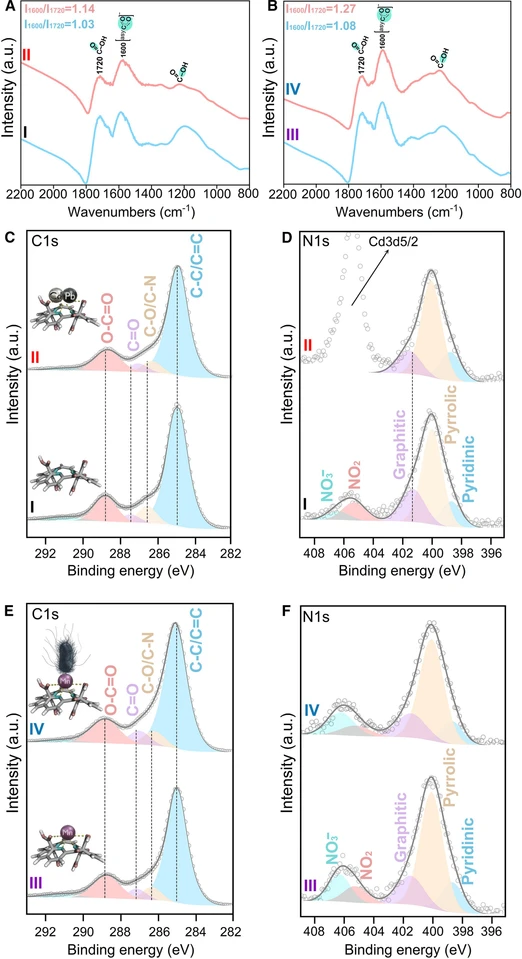 CHEMJOURNAL-D-25-00469: Figure 2. FTIR and XPS characterization (A) FTIR spectra of fresh NGCOOH (I) and of NGCOOH after heavy metals filtration (II). (B) FTIR spectra of fresh Mn-NGCOOH (III) and of Mn-NGCOOH after bacteria filtration (IV). (C and D) Deconvoluted HR-XPS of the C 1s region (C) and of the N 1s region (D) for pristine NGCOOH (I) and for NGCOOH after heavy metals filtration (II). (E and F) Deconvoluted HR-XPS of the C 1s region (E) and of the N 1s region (F) for Mn-NGCOOH (III) and for Mn-NGCOOH after bacteria filtration (IV). The deconvolution details can be found in Tables S1–S6 and the supplemental information.
CHEMJOURNAL-D-25-00469: Figure 2. FTIR and XPS characterization (A) FTIR spectra of fresh NGCOOH (I) and of NGCOOH after heavy metals filtration (II). (B) FTIR spectra of fresh Mn-NGCOOH (III) and of Mn-NGCOOH after bacteria filtration (IV). (C and D) Deconvoluted HR-XPS of the C 1s region (C) and of the N 1s region (D) for pristine NGCOOH (I) and for NGCOOH after heavy metals filtration (II). (E and F) Deconvoluted HR-XPS of the C 1s region (E) and of the N 1s region (F) for Mn-NGCOOH (III) and for Mn-NGCOOH after bacteria filtration (IV). The deconvolution details can be found in Tables S1–S6 and the supplemental information.
The morphology and microstructure of Mn-NGCOOH were analyzed by high-resolution transmission electron microscopy (HR-TEM, Figure 3A), showing that Mn-NGCOOH comprised few-layered graphene flakes with a size of around 2 μm. The elemental mapping (Figures 3B–3F) shows the homogeneous coverage of the flakes by nitrogen and oxygen, corresponding to the spatial distribution of carbon. In particular, the Mn mapping (Figure 3F) shows the absence of dense Mn regions, indicating the absence of Mn-based nanoparticles. The filter membrane was prepared by controlled deposition of NGCOOH and Mn-NGCOOH on the surface of cellulose paper (see the schematic illustration of the final filter membrane in Figure S5 and more details in section “membrane construction” in the supplemental information). The surfaces were investigated by scanning electron microscopy (SEM, Figures 3G–3N), showing the homogeneous and dense coverage of the cellulose paper by the Mn-NGCOOH flakes (Figures 3G and 3H), which are distinctly different from the structure of neat cellulose paper (Figures 3J and 3K), showing a typical fibrous pattern. An optical photograph showing the Mn-NGCOOH membrane can be seen in Figure 3I, also showing macroscopically the homogeneous cellulose paper coverage. The cross section SEM images of the Mn-NGCOOH membrane (Figures 3M and 3N) deposited over the cellulose paper substrate (Figure 3L) showed a coating thickness of 10 μm.
 CHEMJOURNAL-D-25-00469: Figure 3. The morphology and microstructure of Mn-NGCOOH (A) High-angle annular dark-field (HAADF) image of Mn-NGCOOH flake. (B–E) Energy dispersive X-ray spectroscopy (EDS) chemical mapping of Mn-NGCOOH for (B) carbon, (C) oxygen, and combined chemical mapping of (D) carbon and nitrogen and (E) carbon and oxygen. (F) EDS chemical mapping of Mn-NGCOOH for manganese. (G and H) SEM images of top view for Mn-NGCOOH. (I) A detailed photograph showing the Mn-NGCOOH membrane. (J and K) SEM images of top view for cellulose paper substrate. (L–N) Cross section of the (L) cellulose paper substrate, (M) Mn-NGCOOH, and (N) Mn-NGCOOH with higher magnification.
CHEMJOURNAL-D-25-00469: Figure 3. The morphology and microstructure of Mn-NGCOOH (A) High-angle annular dark-field (HAADF) image of Mn-NGCOOH flake. (B–E) Energy dispersive X-ray spectroscopy (EDS) chemical mapping of Mn-NGCOOH for (B) carbon, (C) oxygen, and combined chemical mapping of (D) carbon and nitrogen and (E) carbon and oxygen. (F) EDS chemical mapping of Mn-NGCOOH for manganese. (G and H) SEM images of top view for Mn-NGCOOH. (I) A detailed photograph showing the Mn-NGCOOH membrane. (J and K) SEM images of top view for cellulose paper substrate. (L–N) Cross section of the (L) cellulose paper substrate, (M) Mn-NGCOOH, and (N) Mn-NGCOOH with higher magnification.
Discussion
This study presents the development of a single-atom graphene-based filtration membrane that simultaneously removes bacteria and heavy metals, particularly effectively, from contaminated water. The graphene derivative equipped with carboxyl groups (NGCOOH) and manganese atoms (Mn-NGCOOH) exhibits a high adsorption capacity for heavy metals and bacteria. It has the ability to effectively filter various bacteria in the presence of heavy metals in distilled, tap, and river water, making it suitable for various pollution scenarios. The membrane can be regenerated and reused for at least twenty cycles while maintaining more than 90% of its initial adsorption capacity, thus addressing cost and sustainability issues. It is designed for user-friendly, on-site decontamination even in off-grid areas, eliminating the need for any electrical supply or specialized training, making it practical and cost effective. This study underscores the potential of single-atom engineering in producing advanced materials for environmental remediation and represents a significant step toward highly efficient, sustainable, and user-friendly water treatment solutions. Notably, it brings a previously untapped concept for single-step bacteria and heavy metal removal technologies, opening the door for future advances in filtration materials through their simple modification with benign metal cations.
Materials and Methods
Instrumentation
HR-TEM images were obtained using a HR-TEM TITAN 60-300 microscope with an X-FEG type emission gun, operating at 300 kV. Scanning transmission electron microscopy high-angle annular dark-field imaging (STEM-HAADF) analysis for EDS (energy-dispersive X-ray spectroscopy) elemental mapping on the products was performed with a FEI Titan HR-TEM microscope operating at 80 kV. For this analysis, a droplet of an aqueous dispersion of the material under study with a concentration of ~0.1 mg mL–1 was deposited on a carbon-coated copper grid and dried at room temperature for 24 hours. The filter membrane morphology was examined through scanning electron microscopy (SEM) JEOL 7900F microscope (JEOL, Japan), with an accelerating voltage of 5 kV.
The concentration of Mn in Mn-NGCOOH dispersion was measured in an atomic absorption spectroscopy (AAS), on a ContrAA 600 with graphite furnace (Analytik Jena AG, Germany) equipped with a high-resolution Echelle double monochromator (spectral band width, 2 pm at 200 nm) and a xenon lamp as a continuum radiation source. In order to confirm the measurement, the concentration was further measured using 7500ce inductively coupled plasma mass spectrometry (ICP-MS) instrument (Agilent). 1 mg (weighted on a six decimal digit balance) of the material was added to a 5% nitric acid solution to form a final solution of 50 ml. The solution thus prepared was sonicated for 3 h in order to unbind all Mn ions from the graphene surface. After sonication, the solution was filtered using a 100 nm filter to remove all graphene material from the solution. Subsequently, the filtrate was collected and measured by ICP-MS and AAS to determine the amount of Mn that was bound to Mn-NGCOOH.
FTIR spectra were recorded on an iS5 FTIR spectrometer (Thermo Nicolet) using the Smart Orbit ZnSe ATR accessory. Briefly, a droplet of water dispersion of the relevant material was placed on the ZnSe crystal and dried. The spectra were then acquired by summing 52 scans while using a nitrogen gas flow through the ATR accessory. ATR and baseline correction were applied to the collected spectra.
The XPS measurements were performed using the Nexsa G2 XPS system (Thermo Scientific) equipped with a monochromatic Al Kα X-ray source (hν =1486.68 eV). Spectra acquisition was conducted under ultra-high vacuum conditions (1.0 × 107 Pa) at room temperature (20°C). The analysis area on each sample was a circular spot with a diameter of 300 μm. Survey spectra were recorded with a pass energy of 100.00 eV and a step size of 1.0 eV, while high-resolution spectra were collected with a pass energy of 10.00 eV and a step size of 0.1 eV. During all measurements, a charge neutralisation system was employed to prevent charging effects, ensuring accurate peak positions and reliable quantitative results. Charge neutralisation was achieved by using dual-beam charge compensation (low-energy electrons combined with low-energy ions), effectively neutralising the charge build-up on the surface of the sample. Data acquisition and analysis were performed using Avantage software (version 6.6.1). For peak fitting, a mixed Lorentzian/Gaussian line shape with a 30% Lorentzian contribution was used, representing a common compromise between instrumental broadening and natural line width, except the C–C (sp 2 ) peak was fitted using an asymmetric line shape due to the conductive nature of the graphitic material. For background subtraction, the so-called Smart Background algorithm implemented in Anvantage software was used, which is based on the Shirley method with the additional constraint that the background intensity cannot exceed the measured signal.