Enhancing Paper’s Mechanical Strength and Antibacterial Properties through a Biopolymer-Based Coating
Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Graphical abstract
This study demonstrates a sustainable method to enhance paper’s strength and antibacterial properties using a biopolymer coating derived from citric acid–modified soybean flour (SBFC) and carboxymethyl cellulose (CMC). Optimized at a 50:50 ratio, the coating achieved a 61.8% increase in tensile strength compared to uncoated paper.
Antibacterial performance was evaluated against Escherichia coli, with results showing an 84–89% reduction in viable colonies. While tested on a single strain, these findings highlight the potential of SBFC/CMC coatings as multifunctional additives for improving both durability and hygiene in paper packaging applications.
The original article
Enhancing Paper’s Mechanical Strength and Antibacterial Properties through a Biopolymer-Based Coating
Neha Sawant, Sara T. Caceres, Carin L. Garcia, Mario O. C. Lizardo, Carol Beaver, Santiago Aparicio, and Mert Atilhan*
Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013
https://doi.org/10.1021/acs.iecr.5c01067
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
The growing demand for paper-based packaging as a sustainable alternative to petroleum-derived plastics faces challenges due to paper’s natural hydrophilicity and lack of antimicrobial properties. To address these limitations, this study presents a simple, eco-friendly approach to enhance paper with antimicrobial and strength functionalities, making it suitable for diverse packaging applications.
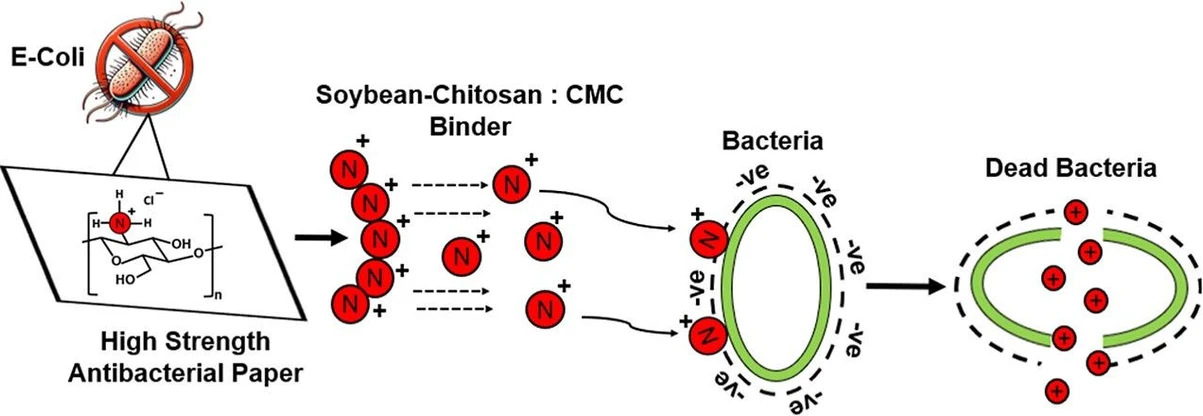
Chitosan, a biocompatible and biodegradable polysaccharide derived from chitin, has emerged as a promising material for drug delivery and biomedical applications. Its unique properties, influenced by various extraction techniques, can be further enhanced through chemical modifications. (1) The presence of reactive hydroxyl and amino groups facilitates intramolecular and intermolecular hydrogen bonding, contributing to chitosan’s nontoxic, antibacterial, antioxidative, and immunomodulatory properties. Cationic polysaccharides with amino or ammonium groups are valuable in various applications, and their chemical modification has been extensively studied to create anionic, cationic, and amphoteric materials for use in paper, textiles, and food industries. (2) Chitosan shows a broad spectrum of antibacterial activity against both Gram-negative and Gram-positive bacteria, though its effectiveness varies across species. (3) Chitosan’s antibacterial action is believed to result from a series of untargeted molecular events, including electrostatic interactions with teichoic acids on the bacterial cell wall, membrane disruption causing leakage of cellular components, interference with membrane-bound energy pathways, and potential stress responses due to local pH changes. (4) Chitosan’s antimicrobial activity against Escherichia coli and S. aureus was investigated, finding that higher-molecular-weight chitosan forms a surface barrier blocking nutrient intake, while lower-molecular-weight chitosan permeates the cell, causing flocculation and physiological disruption; the former mechanism was seen better in S. aureus, than in E. coli, with 100% inhibition observed at a 1% chitosan concentration. (5) A study showed an eco-friendly method for enhancing cellulose paper with antimicrobial and hydrophobic properties through chemical grafting of 3-aminopropyltriethoxysilane (APS) and cinnamaldehyde (CA), resulting in paper with long-term antibacterial and antifungal effects, good mechanical strength even when wet, excellent biocompatibility and biodegradability, and improved shelf life for strawberries, making it a promising material for fruit packaging. (6) Biopolymer nanoparticles form a unique biobased latex emulsion that serves as an alternative binder to petrochemical-based systems in coated paper and paperboard manufacturing, with re-engineered starch nanoparticles that form a colloidal dispersion in water; these binders, when combined with titanium dioxide through reactive extrusion, enhance the optical properties of the coated paper by improving whiteness and brightness.
The imperative to mitigate the environmental burden associated with petroleum-based plastics has catalyzed significant research on sustainable packaging alternatives. Biopolymers, sourced from renewable biological origins, represent a highly promising class of materials owing to their inherent biodegradability, biocompatibility, and independence from fossil fuel feedstocks. (7) Chitosan and soy protein, in conjunction with modified cellulosic materials, such as carboxymethyl cellulose (CMC), exemplify this potential. Soy protein, economically derived from abundant agricultural sources, exhibits favorable film-forming capabilities. (8−10) Chitosan, the deacetylated derivative of chitin (a structural polysaccharide abundant in crustacean shells, insect exoskeletons, and fungal cell walls), offers unique antimicrobial properties alongside its biodegradability and nontoxicity. (11)
The antimicrobial action of chitosan is multifaceted yet fundamentally rooted in its polycationic character under acidic conditions. The positively charged amino groups (NH3+) present along the chitosan polymer chain engage in electrostatic interactions with negatively charged constituents of microbial cell membranes, such as lipopolysaccharides in Gram-negative bacteria and teichoic acids in Gram-positive bacteria. (12,13) These interactions compromise membrane integrity, inducing leakage of intracellular components (proteins, nucleic acids, and ions), ultimately culminating in cell death. (14) The degree of deacetylation (DDA) and molecular weight of chitosan are critical determinants of its antimicrobial efficacy. A higher DDA, reflecting a greater proportion of free amino groups, generally enhances antimicrobial potency due to increased positive charge density. (15) The relationship between MW and antimicrobial activity is more nuanced; higher-molecular-weight chitosan may create a physical barrier impeding nutrient uptake, while lower-molecular-weight chitosan can penetrate the cell membrane, disrupting intracellular processes. (14)
Recent research has also emphasized the synergistic effects of combining chitosan with other plant-based proteins such as soy flour, highlighting improvements in film-forming capacity, water resistance, and antibacterial activity. (16,17) Furthermore, soy protein has been studied extensively for its adhesive properties, and its functional groups can interact with chitosan to provide enhanced bonding and antimicrobial characteristics. (16) Citric acid cross-linking is recognized as an effective, nontoxic strategy for stabilizing biopolymer-based coatings, as it can form ester linkages that improve thermal stability and moisture resistance. (17) Additionally, blending chitosan with carboxymethyl cellulose (CMC) has been shown to strengthen the mechanical properties of coatings while retaining desirable antibacterial effects. (18) In this study, citric acid was chosen as the modifying agent to optimize the reaction conditions and enhance antimicrobial activity. Citric acid was reacted with soy flour in the presence of sodium hypophosphite and then complexed with chitosan. A fixed amount of the modified soy flour additive, along with a 1% CMC solution slurry, was uniformly applied to hand sheets for all tests. (19)
2. Materials and Methods
2.4. Characterization
2.4.1. FTIR Analysis
Fourier transform infrared (FTIR) spectroscopy was used to characterize the functional groups in soybean flour before and after modification. The analysis was conducted using a PerkinElmer Spectrum 100 FTIR spectrophotometer with an attenuated total reflectance (ATR) accessory. (25) Spectra were recorded in the range of 4000–600 cm–1 with background correction performed before each measurement to eliminate atmospheric interference. (26) A small amount of soybean flour and modified soybean flour was placed individually on the ATR crystal, and uniform pressure was applied using the built-in pressure clamp to ensure proper contact. The FTIR spectra, collected in absorbance mode, provided details on structural modifications such as the esterification reaction. This analysis was essential for evaluating changes in functional groups, to identify and confirm the effectiveness of the modification process. (27,28)
2.4.2. Titration Procedure
The carboxylic content of the modified soybean flour was determined using an acid–base titration. 0.5 g of each soybean flour sample was weighed and transferred into 250 mL Erlenmeyer flasks. 20 mL of a 0.1N NaOH solution was added to each flask using a pipet. The flasks were placed on a shaker and mixed for 30 min to allow thorough dispersion of the flour samples in the NaOH solution. Figure 1 shows the sample preparation for titration. (28)
 Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 1. Sample preparation for titration.
Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 1. Sample preparation for titration.
2.4.3. Scanning Electron Microscopy
A scanning electron microscope (SEM) operates similarly to an optical microscope but uses a focused beam of electrons instead of light to capture images and analyze a specimen’s structure and composition. In this study, the surface morphology of the control handsheet, SBFC/CMC-coated handsheets, and SBFC-coated new sheets was analyzed by using a JEOL JSM-IT200 scanning electron microscope (SEM). To enhance the conductivity and improve image contrast, a thin layer of gold was sputter-coated onto the samples under vacuum conditions using a DII-29010SCTR Smart Coater before imaging. The electron source generates a beam that is accelerated toward the specimen by using a positive electrical potential. As the electrons interact with the atoms in the sample, they generate signals that provide insight into surface topography, composition, and electrical properties. These signals are then processed and converted into detailed images of the specimen. (28,29)
2.4.7. Dynamic Viscosity Measurement of the 50:50 SBFC/CMC Solution
The dynamic viscosity of the 50:50 SBFC/CMC (vol: vol) solution was measured using an Anton Paar ViscoQC300 instrument (Figure 5).
3. Results and Discussions
The esterification reaction for modifying defatted soybean flour with citric acid was carried out at 120 °C for 2 h. The reaction time was carefully monitored and started only after the mixture reached the desired temperature. Following the completion of the reaction period, images of the samples were taken to document visual differences in color and appearance, confirmed with varying concentrations of citric acid used (20%, 40%, 60%, 80%, and 100%); Figure 8. Each formulation is coded as CxSy to indicate the specific citric acid-to-soybean flour ratio used during synthesis.
 Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 8. Effect of citric acid concentration on soybean flour (before the washing step).
Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 8. Effect of citric acid concentration on soybean flour (before the washing step).
These pictures indicate the effect of the citric acid concentration on the modification process before the washing and purification steps.
3.1. Scanning Electron Microscopy (SEM)
The optimized soybean flour was analyzed with SEM and FTIR spectroscopy. The surface of soybean flour went through significant changes after modification with citric acid at 140 °C for 2 h, as observed through SEM. The SEM images (Figure 15) showed that the untreated soybean flour initially had a smooth and uniform surface morphology. Green dashed circles highlight areas of increased surface roughness observed in the citric acid–modified soybean flour, while yellow dashed circles indicate regions where particles appear more compact and fused, suggesting enhanced structural cohesion resulting from cross-linking.
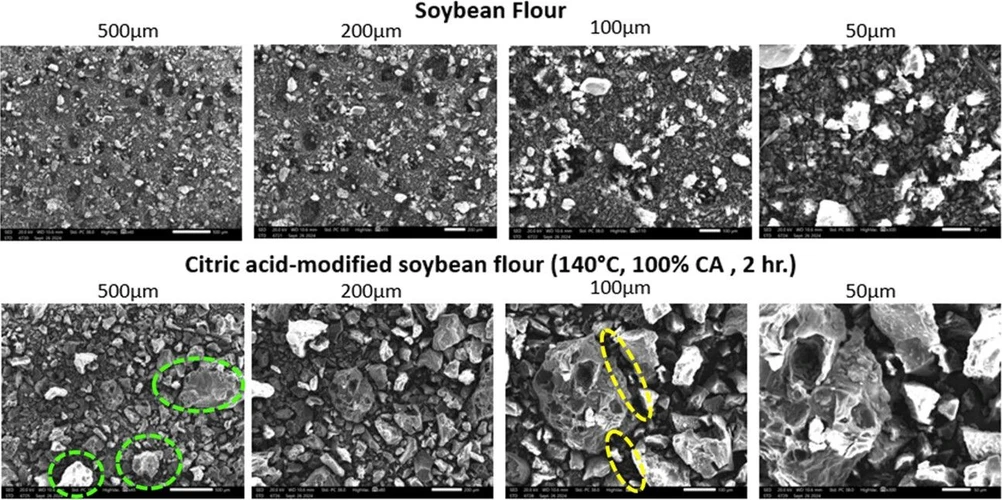 Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 15. SEM of soybean flour vs citric acid-modified flour.
Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 15. SEM of soybean flour vs citric acid-modified flour.
However, after modification, the surface became rougher and coarser. This could be due to the esterification reaction between the hydroxyl groups of the soybean flour and the carboxylic groups of citric acid, which led to an increased carboxylic content. The chemical modification process could have added to the structural changes in the flour particles; this could be due to cross-linking and degradation due to the high temperature and long reaction time. Hence, these changes suggest successful functionalization.
3.3. FTIR Spectroscopy
The FTIR spectra of unmodified soybean flour and citric acid-modified soybean flour show changes from the esterification reaction (Figure 18). The unmodified soybean flour showed peaks at 3273, 1635, 1531, 1393, 1237, 1047, and 998 cm–1. The peak at 1635 cm–1 indicates C═O stretching of amide bonds, which is a prominent indicator of proteins. (33) The peak at 1531 cm–1 represents N–H bending and C–N stretching vibrations. (34) The peaks at 1237 and 1047 cm–1 are due to the C–O stretching in carbohydrates, and the peak at 998 cm–1 corresponds to C–H bending.
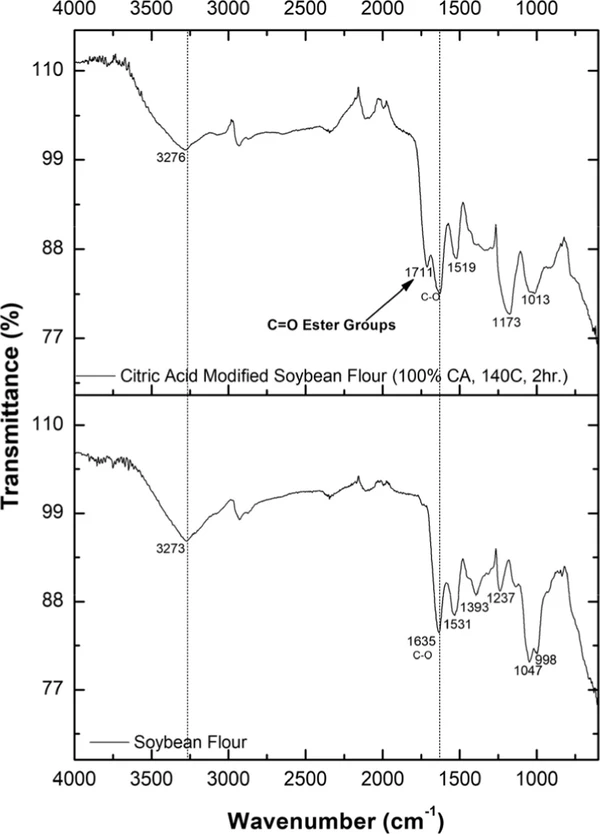 Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 18. FTIR spectra of unmodified soybean flour and citric acid-modified soybean flour.
Ind. Eng. Chem. Res. 2025, 64, 24, 12000–12013: Figure 18. FTIR spectra of unmodified soybean flour and citric acid-modified soybean flour.
4. Conclusion
The study focused on improving the mechanical and antibacterial properties of the paper. The formulation was optimized by using a citric acid modification process for soybean flour, resulting in an increase in carboxyl content. Final optimized conditions were 140 °C for 2 h with 100% citric acid and 15% Na2HPO4. FTIR spectra proved successful esterification in citric acid-modified soybean flour, with a new ester carbonyl peak at 1711 cm–1. Citric acid-modified soybean flour was further complexed with chitosan. Further, a 50:50 vol: vol SBFC/CMC solution was optimized and used as a coating material for handsheets.
Surface morphology analysis through SEM confirmed that the coating effectively filled gaps between fibers, creating a denser structure compared with the control handsheets. Tensile strength testing, following TAPPI T494 standards, showed that the coated handsheets had a significant improvement in tensile strength (1.552 kgf) compared to the control handsheets (0.959 kgf).
Both antibacterial treatments, SBFC-only coating and SBFC/CMC (50:50 vol: vol) double-sided coating, showed significant bacterial reduction. The SBFC-only coated new sheet showed complete antibacterial activity, resulting in a 100% reduction in bacterial colonies, whereas the double-sided 50:50 vol: vol SBFC/CMC-coated handsheet showed an 84.6% reduction compared to the control E. coli sample and at least an 89.5% reduction compared to the untreated handsheet. Additionally, antibacterial testing at OD600 values of 0.104, 0.138, and 0.157 showed consistent reductions in bacterial colony counts across 3 trials, with the coated handsheet reducing growth by up to 80% compared to control E. coli. These results suggest that the SBFC-only coating is more effective in eliminating bacterial colonies entirely; the SBFC/CMC coating still provides substantial bacterial reduction and also improves the mechanical strength of the handsheet. If antibacterial efficiency is the priority, the SBFC-only coating is superior due to its complete bacterial inhibition. However, if a balance between antibacterial action and mechanical strength is needed, the SBFC/CMC coating remains a strong alternative, offering high bacterial reduction along with good strength.
While the CFU plate count method used here aligns with accepted standards such as ISO 22196, we acknowledge its limitations, including the inability to detect viable but nonculturable (VBNC) cells. Future work should incorporate complementary techniques, such as Live/Dead fluorescence staining or SEM-based membrane integrity assessment, to confirm bactericidal activity.