PCPDTBT Conjugated Polymer Nanoparticles for Photothermal Inactivation of Multidrug-Resistant mcr-1-Positive Escherichia coli
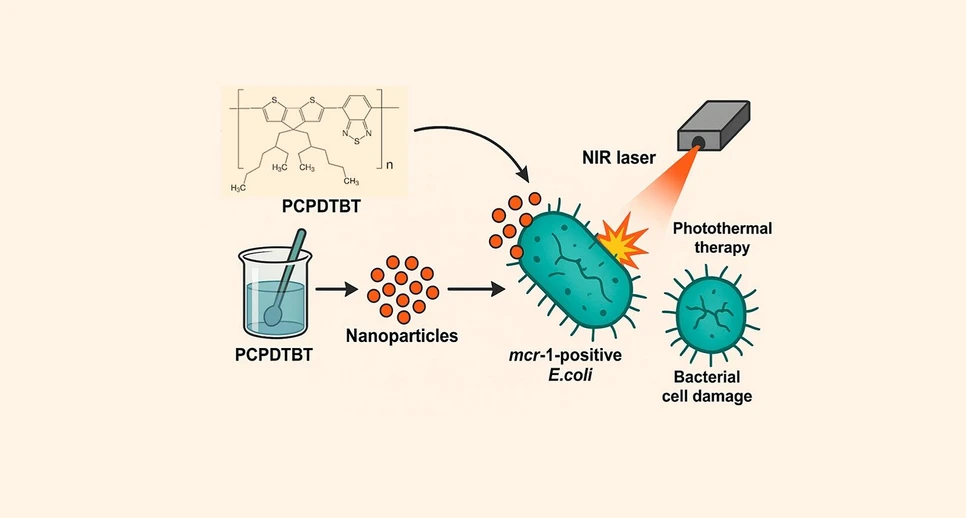
ACS Omega 2025, 10, 35, 40111–40118: Graphical abstract
Antimicrobial resistance, especially plasmid-mediated mcr-1 in E. coli, limits the effectiveness of last-resort antibiotics such as colistin. This study evaluates conjugated PCPDTBT polymer nanoparticles as photothermal agents for combating these multidrug-resistant strains.
The nanoparticles were produced by nanoprecipitation, showed strong NIR absorption, and generated substantial heat under 806 nm irradiation. They remained photothermally stable across repeated cycles and significantly reduced the viability of mcr-1-positive E. coli. SEM imaging confirmed severe structural damage after treatment. These findings demonstrate the strong potential of PCPDTBT nanoparticles as standalone photothermal therapeutics against antibiotic-resistant pathogens.
The original article
PCPDTBT Conjugated Polymer Nanoparticles for Photothermal Inactivation of Multidrug-Resistant mcr-1-Positive Escherichia coli
Cynthia S. A. Caires, Rafael C. Nascimento, Leandro O. Araujo, Laís F. Aguilera, Samuel L. Oliveira*, and Anderson R. L. Caires*
ACS Omega 2025, 10, 35, 40111–40118
https://doi.org/10.1021/acsomega.5c04993
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
The World Health Organization (WHO) has identified antimicrobial resistance (AMR) as one of the top 10 global public health threats. (1) AMR in bacterial pathogens is a growing pandemic that threatens to render current treatments for microbial infections ineffective, bringing back a preantibiotic era. In 2019, approximately 1.27 million people worldwide died from infections directly caused by antibiotic-resistant bacteria. (2,3) Without innovative interventions, global deaths associated with AMR are projected to reach 10 million annually by 2050. (4)
Conjugated polymer nanoparticles (CPNs) are multifunctional nanoscale materials that exhibit versatile applications across various technological fields, drawing attention for their use in photothermal and photodynamic strategies to combat multidrug-resistant bacteria. (19−23) The photothermal approach can be achieved by combining the use of CPNs under near-infrared (NIR) illumination, (24) in which CPNs act as photothermal agents (PTAs) because they efficiently convert NIR light into heat, resulting in heat-induced bacterial death promoted by localized hyperthermia. (25,26) This method offers a novel, noninvasive approach to tackling antibiotic resistance, reducing the dependence on conventional treatments, (22,25) whose effectiveness has already been demonstrated in vitro and in vivo. (18,27)
Poly[2,6-(4,4-bis(2-ethylhexyl)-4H-cyclopenta[2,1-b;3,4-b′]dithiophene)-alt-4,7(2,1,3-benzothiadiazole)], also known as PCPDTBT, is a conjugated polymer with luminescent properties that has also been studied as a PTA under NIR light, acting as a theranostic agent for photothermal and imaging applications. (26,28,29) MacNeill et al. (2013) demonstrated that PCPDTBT nanoparticles can effectively generate localized heat upon NIR irradiation, enabling thermal ablation of cancer cells in vitro. (30) Subsequent study has highlighted how molecular ordering within nanoparticle assemblies significantly influences their photothermal performance. (31) Additionally, PCPDTBT-based systems have been integrated into multifunctional nanoplatforms for synergistic therapeutic strategies. For instance, Zhou et al. (2022) developed chemiluminescent nanoprobes incorporating PCPDTBT for deep-tissue bacterial imaging and combined photothermal-nitric oxide therapy. (32) More recently, Wu et al. (2025) demonstrated that NIR-responsive polymer dots containing PCPDTBT can enhance the synergistic effect of photothermal therapies in vitro. (29) Together, these studies underscore PCPDTBT’s versatility and efficacy as a core photothermal material in biomedical applications, with significant potential for in vivo applications due to its established biocompatibility and biodegradability. (33,34) However, long-term in vivo studies of clearance mechanisms and immune responses remain an area for further investigation to fully validate its clinical translational potential.
Although PCPDTBT has been investigated as a PTA, its photothermal action against multidrug-resistant bacteria remains unexplored. The present study shows that PCPDTBT NPs produced by the nanoprecipitation method efficiently photoinactivate the mcr-1 E. coli strain after a few minutes of NIR irradiation.
2. Materials and Methods
2.1. Nanoparticle Synthesis
Conjugated polymer poly[2,6-(4,4-bis(2-ethylhexyl)-4H-cyclopenta[2,1-b;3,4-b′]dithiophene)-alt-4,7(2,1,3-benzothiadiazole)] (PCPDTBT) from Sigma-Aldrich was used to produce nanoparticles by nanoprecipitation. (15) PCPDTBT was added dropwise to tetrahydrofuran (THF) at 0.23 mg mL–1 to 10 mL of an aqueous solution of polysorbate 20 (Tween 20, Quimesp Química, Brazil) at 1.2 mg mL–1 under slow stirring. After the mixture was stirred for 12 h, the evaporated volumes of water and THF were refilled with the surfactant solution to maintain the nanosuspension volume at 10 mL (Figure 1a). The final concentration of PCPDTBT NPs in the stock solution was 55.3 mg L –1.
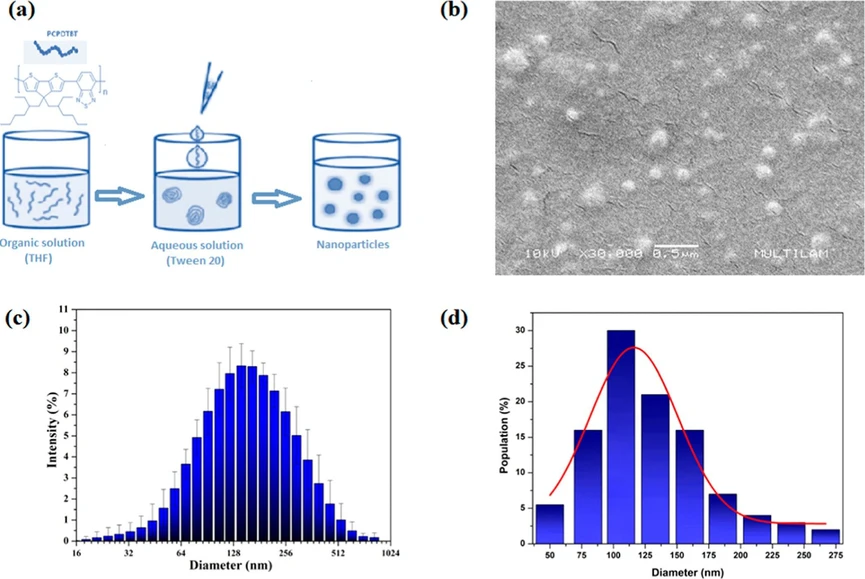 ACS Omega 2025, 10, 35, 40111–40118: Figure 1. (a) Schematic representation of the nanoprecipitation process; (b) scanning electron microscopy (SEM) image of PCPDTBT nanoparticles; (c) hydrodynamic diameter distribution of PCPDTBT NPs; (d) particle diameter distribution determined from SEM image analysis.
ACS Omega 2025, 10, 35, 40111–40118: Figure 1. (a) Schematic representation of the nanoprecipitation process; (b) scanning electron microscopy (SEM) image of PCPDTBT nanoparticles; (c) hydrodynamic diameter distribution of PCPDTBT NPs; (d) particle diameter distribution determined from SEM image analysis.
2.2. Characterization
2.2.1. UV–vis–NIR Absorption
UV–vis–NIR spectroscopy was carried out using a Shimadzu UV-2600i spectrophotometer equipped with an ISR-2600 Plus integrating sphere accessory, which collected the absorption spectrum in the range of 300–900 nm.
2.2.2. Size and Morphology
The size and morphology of PCPDTBT NPs were analyzed by using scanning electron microscopy (SEM) (JEOL model JSM-6380LV) at an operating voltage of 10 kV. A solution of PCPDTBT NPs was applied onto a 1 cm × 1 cm glass substrate and allowed to dry overnight at room temperature. The dried sample was sputter-coated with gold and attached to SEM holders with carbon. ImageJ software was used to analyze SEM images to determine the particle diameter distribution and morphology. The average size was calculated by assuming a spherical shape for 100 particles. The hydrodynamic size, polydispersity index, and zeta potential were measured using dynamic light scattering (DLS) with a Zetasizer NanoZS device from Malvern Instruments Ltd., UK. The mean hydrodynamic diameter of PCPDTBT NPs was measured at 25 °C immediately after diluting to a concentration of 4.9 μg mL–1. NaCl was added to the PCPDTBT NP solutions to achieve a final concentration of 9 mM for the zeta potential measurements.
3. Results and Discussion
3.2. Optical and Thermal Characterization
Figure 2b shows broad absorption bands of the solution with PCPDTBT NPs (17.0 mg L–1) from the UV to the near-infrared range (350–900 nm). Abelha et al. (2019) previously studied PCPDTBT NPs for their potential as fluorescent agents in assessing near-infrared wavelengths in animal systems. (34,41) The study also examined their cytotoxicity and hemocompatibility, particularly their compatibility with animal blood. The findings indicated that PCPDTBT NPs effectively produced near-infrared images, demonstrated low cytotoxicity, and exhibited high hemocompatibility, establishing them as reliable and biocompatible materials for various biomedical applications. (34)
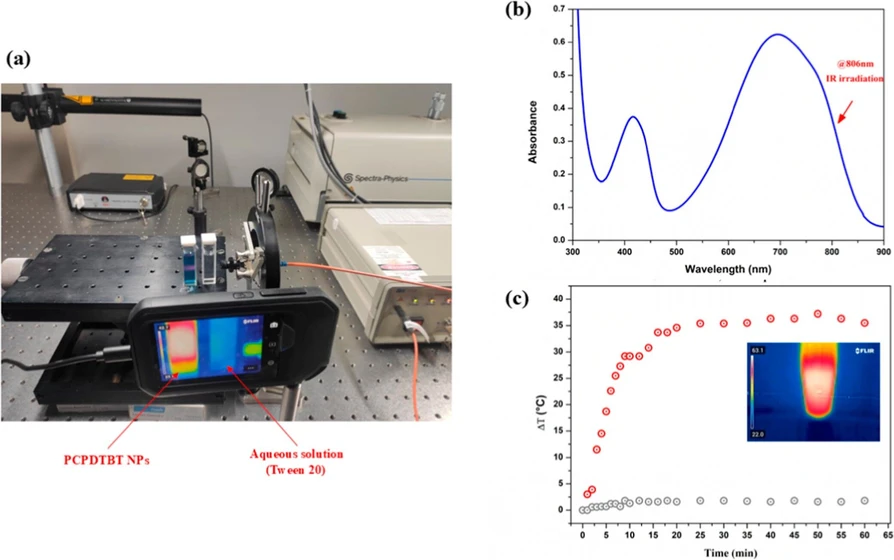 ACS Omega 2025, 10, 35, 40111–40118: Figure 2. (a) Experimental setup for photothermal analysis; (b) absorption spectrum of PCPDTBT NPs (17.0 mg L–1). The arrow indicates the absorbance peak at 806 nm; (c) temperature as a function of time upon near-infrared irradiation (806 nm) for PCPDTBT nanoparticles (17.0 mg L–1) dispersed in aqueous solution and aqueous solution with Tween 20.
ACS Omega 2025, 10, 35, 40111–40118: Figure 2. (a) Experimental setup for photothermal analysis; (b) absorption spectrum of PCPDTBT NPs (17.0 mg L–1). The arrow indicates the absorbance peak at 806 nm; (c) temperature as a function of time upon near-infrared irradiation (806 nm) for PCPDTBT nanoparticles (17.0 mg L–1) dispersed in aqueous solution and aqueous solution with Tween 20.
Several works use different matrices for the encapsulation of NPs and also associate them with other materials such as PCPDTBT; it was encapsulated with a layer of Pluronic F127 amphiphilic block and irradiated with a NIR laser at 785 nm, and the nanoparticles obtained present good thermal response and excellent photostability, leading to a photothermal conversion efficiency of 61%. (40)
3.3. Photothermal Response and Stability
It was possible to observe that the PCPDTBT NPs produced in Figures 3a and 3c require an adequate concentration and potency to obtain their peak efficiency, achieving a maximum temperature increase (ΔT) (°C) of 35–40 °C over time under infrared irradiation (806 nm) at different laser powers (0.22, 0.52, 0.81, and 1.13 W) at the concentration of 17.0 mg L–1 for PCPDTBT NPs and various concentrations of PCPDTBT NPs (0, 4.25, 8.50, and 17.0 mg L–1) a function of the time under infrared irradiation. Figures 3b and 3d also show the linear increase in temperature as a function of power and concentration, respectively.
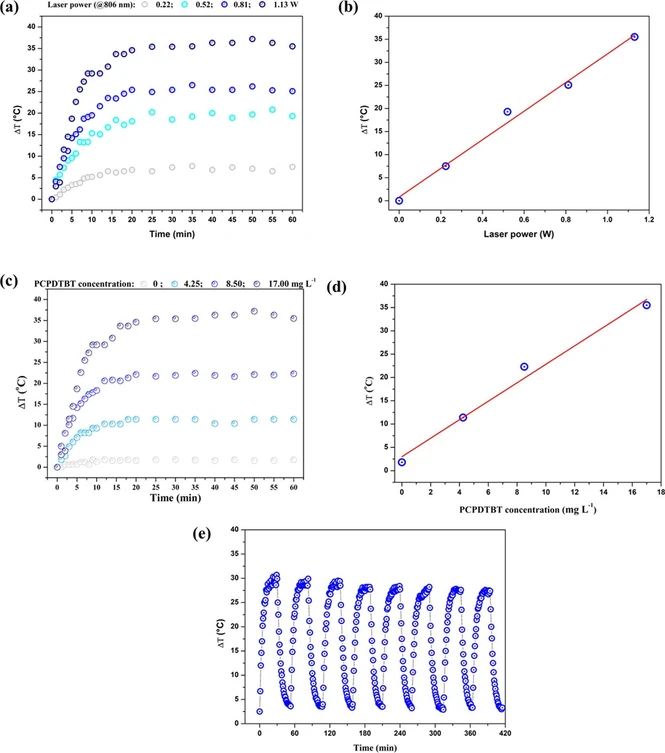 ACS Omega 2025, 10, 35, 40111–40118: Figure 3. (a) Temperature of PCPDTBT NP solutions (17.0 mg L–1) as a function of time under 806 nm irradiation at laser powers (0.22, 0.52, 0.81, and 1.13 W); (b) solution temperature after 60 min of irradiation as a function of PCPDTBT NP concentration at 1.13 W (data from (a)); (c) temperature as a function of time of PCPDTBT NPs at different concentrations (0, 4.25, 8.50, and 17.0 mg L–1), irradiated at 806 nm irradiation at 1.13 W; (d) temperature of solutions after 60 min of irradiation extracted from data presented in (c); (e) cyclic photothermal heating of PCPDTBT NP solution (17.0 mg L–1) irradiated at 806 nm with 1.13 W over eight heating–cooling cycles.
ACS Omega 2025, 10, 35, 40111–40118: Figure 3. (a) Temperature of PCPDTBT NP solutions (17.0 mg L–1) as a function of time under 806 nm irradiation at laser powers (0.22, 0.52, 0.81, and 1.13 W); (b) solution temperature after 60 min of irradiation as a function of PCPDTBT NP concentration at 1.13 W (data from (a)); (c) temperature as a function of time of PCPDTBT NPs at different concentrations (0, 4.25, 8.50, and 17.0 mg L–1), irradiated at 806 nm irradiation at 1.13 W; (d) temperature of solutions after 60 min of irradiation extracted from data presented in (c); (e) cyclic photothermal heating of PCPDTBT NP solution (17.0 mg L–1) irradiated at 806 nm with 1.13 W over eight heating–cooling cycles.
Figure 3e demonstrates the thermal stability of PCPDTBT NPs at 17.0 mg L–1 even after 8 cycles of 60 min of irradiation at 1.13 W.
The PCPDTBT NPs have gained widespread application in the biomedical field since they have been used strictly as fluorescent markers (42,43) and photoresponsive agents for heat generation, often in conjunction with other agents. (30) The PCPDTBT-containing hybrid nanoparticles were also explored for the aPDI properties, acting as coagents for diagnostic imaging or photothermal therapy, used in association with other agents. (21,44,45) Despite the existence of these works, this report presents, for the first time, the idea that nanoparticles composed solely of PCPDTBT can be utilized as a photothermal agent to inactivate multidrug-resistant bacteria.
4. Conclusion
In conclusion, PCPDTBT NPs are effective photothermal agents against the antibiotic-resistant mcr-1 E. coli strain. The PCPDTBT NPs synthesized via nanoprecipitation exhibited robust photothermal properties characterized by efficient NIR light absorption and subsequent conversion into heat. This capability was validated through a consistent thermal performance over multiple irradiation cycles. Moreover, the photothermal efficacy of PCPDTBT NPs was confirmed through photoinactivation assays, revealing significant reductions in the number of viable bacterial counts after NIR laser treatment. The mcr-1-positive E. coli strain showed notable bactericidal effects, as illustrated by SEM images that revealed significant damage to bacterial morphology following photothermal treatment therapy. These findings highlight PCPDTBT NPs as promising candidates for combating antibiotic-resistant bacteria, offering a low-toxicity alternative to conventional antimicrobial treatments.