UA-DMSPE Determination of Cu(II), Cd(II), and As(III) in Water, Soil, and Tomato Using a Novel Thiosemicarbazone Sorbent: ICP-OES Performance with DFT Characterization and Antimicrobial-Target Docking
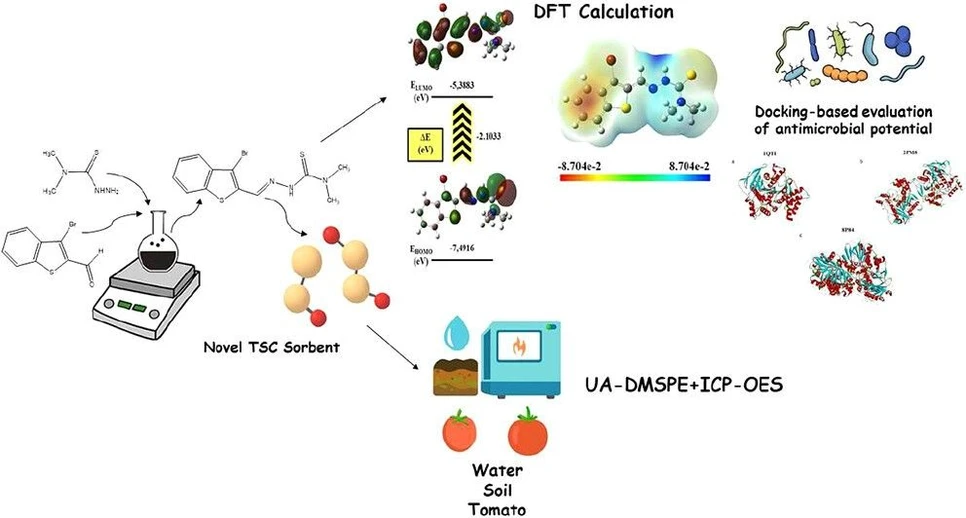
ACS Omega 2026, 11, 1, 1213–1235: Graphical abstract
This study presents a novel thiosemicarbazone-functionalized sorbent for ultrasound-assisted dispersive microsolid phase extraction of Cu(II), Cd(II), and As(III) from environmental and food samples. The method, coupled with ICP-OES, demonstrated high sensitivity, low detection limits, and reliable performance in irrigation water, soil, and tomato matrices.
Density functional theory calculations and molecular docking analyses further explored the ligand’s electronic properties and potential antimicrobial activity. The integrated approach highlights the multifunctional applicability of the sorbent for trace metal monitoring and prospective biomedical research.
The original article
UA-DMSPE Determination of Cu(II), Cd(II), and As(III) in Water, Soil, and Tomato Using a Novel Thiosemicarbazone Sorbent: ICP-OES Performance with DFT Characterization and Antimicrobial-Target Docking
Serkan Öncüoğlu*
ACS Omega 2026, 11, 1, 1213–1235
https://doi.org/10.1021/acsomega.5c08559
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
Heavy metals such as Cu(II), Cd(II), and As(III) pose significant threats to both the environment and human health due to their toxicity, bioaccumulation potential, and persistence in ecosystems. Especially cadmium and arsenic, even at trace concentrations, are known to exert nephrotoxic, neurotoxic, and carcinogenic effects through long-term exposure. (1−6) Therefore, their accurate, sensitive, and selective determination in natural and drinking water sources is one of the critical challenges in environmental monitoring. Although spectroscopic techniques such as ICP-OES and ICP-MS offer high sensitivity, the ultratrace levels of these metals in complex environmental matrices necessitate effective preconcentration strategies to ensure reliable quantification. (7−10) Among various sample preparation techniques, dispersive microsolid-phase extraction (DMSPE) has gained considerable attention due to its rapid phase interaction, low solvent consumption, and elimination of elution steps. (11−21)
In addition to aquatic matrices, the accumulation of heavy metals such as Cu(II), Cd(II), and As(III) in soil poses serious ecological and agricultural threats. (22−24) However, the accurate determination of heavy metals in soils is complicated by strong adsorption to heterogeneous mineral phases, binding to humic substances, and site-specific variability, all of which can obscure true analyte concentrations and hinder reproducibility. Vegetables cultivated in metal-contaminated soils such as tomatoes can absorb these elements and introduce them into the human food chain either directly or through animal-derived products. This exposure route may result in severe health effects, including gastrointestinal and respiratory disorders, liver toxicity, and even cancer. (25−27) Moreover, the trace metal balance in soil, including essential elements such as Zn, Fe, and Cu, significantly affects plant health and crop productivity. While micronutrients are vital for growth, excessive levels of certain metals (e.g., Al, Fe) may negatively impact soil fertility. (28−32) Hence, trace metal analysis in soil is crucial not only for toxicological assessment but also for sustainable agricultural practices. (33−35)
Plant-based matrices, particularly edible vegetables, are also valuable indicators of environmental contamination. Nevertheless, accurate monitoring in plant tissues remains challenging due to multistep pretreatment requirements (washing, drying, grinding, digestion) and the high organic content of the matrix, which can cause spectral interferences and analyte loss during preparation. Major elements like Ca, K, and Mg, along with trace essential elements such as Fe, Zn, Mn, Cu, and Se, are vital for human metabolism. (36−39) However, their excess or deficiency may lead to various disorders, and toxic elements such as Cd, Pb, As, and Hg can pose serious health risks even at very low concentrations. (40−44) Inductively coupled plasma optical emission spectroscopy (ICP-OES) remains a leading technique for elemental analysis in plant samples due to its multielement detection, broad linear range, and low detection limits. (45−49) Yet, accurate analysis of solid plant matrices like vegetables requires multistep sample preparation including washing, drying, grinding, acid digestion, and filtration before instrumental measurement. (50) In line with the principles of green analytical chemistry, recent efforts focus on minimizing reagent use, simplifying procedures, and improving environmental sustainability in such analytical workflows. (51−54)
A critical factor in DMSPE is the selection of a suitable chelating agent that offers fast complexation, aqueous solubility, and stability over a wide pH range. In this context, thiosemicarbazones (TSCs) have emerged as highly effective ligands due to their polydentate nature and versatile coordination behavior, especially toward soft metal ions such as Cu(II), Cd(II), and As(III). (55−65) Literature reveals that TSC ligands not only improve analytical performance in metal extraction but also display significant biological activities including antibacterial, antifungal, antituberculosis, and anticancer properties. (66−68) Moreover, the incorporation of nitrogen-containing heterocycles, such as pyrazole rings, into TSC structures has been shown to enhance both coordination capacity and pharmacological potential. (69) These structural features increase cell membrane permeability and biological target interaction, broadening their application in medicinal chemistry. (70−76) TSC-based metal complexes, particularly with Cu(II), Ni(II), Co(II), Pt(II), and Ru(II), have exhibited promising results against various bacterial strains and tumor cell lines. (77−79)
Recent studies have also demonstrated the potential of nanoparticle-functionalized sorbents such as Fe3O4 magnetic cores coated with poly(8-hydroxyquinoline) for the selective and efficient extraction of Cu(II) from complex matrices like soil, tomato, and water samples. (80) These materials offer excellent surface area, tunable selectivity, and low detection limits, while their magnetic responsiveness facilitates rapid separation without centrifugation. (81−84) The selection of water, soil, and tomato matrices was made to encompass distinct yet complementary exposure pathways: water as a direct route for both human consumption and agricultural applications, soil as the principal environmental reservoir and source of contamination, and tomato as a representative edible crop widely consumed for its capacity to accumulate trace metals from contaminated soils.
In this study, I report the synthesis and structural characterization of a novel thiosemicarbazone derivative not previously described in the literature. The synthesized ligand was covalently immobilized onto a silica-based sorbent and applied in the UA-DMSPE procedure for the selective extraction and enrichment of Cu(II), Cd(II), and As(III) from environmental water, soil, and tomato samples, followed by quantification via ICP-OES. In this regard, the UA-DMSPE approach developed in this study minimizes sample handling steps, reduces matrix interferences, and enhances recovery efficiency, thereby addressing many of the limitations commonly associated with soil- and plant-based matrices. The novelty of this work lies in combining a newly designed TSC derivative with UA-DMSPE to achieve a high recovery at trace levels with improved reproducibility. Furthermore, the integration of DFT calculations and docking studies provides a unique interdisciplinary perspective, reinforcing both the environmental monitoring applications and the potential biomedical relevance of the ligand.
Experimental Section
The structural characterization of the synthesized compound was carried out using 1H NMR spectroscopy on a high-resolution Bruker WH-400 Fourier Transform NMR spectrometer. FTIR spectra were recorded by using a PerkinElmer Spectrum BX-II instrument. Elemental compositions (C, H, N, and S) were determined by using a LECO CHNS-O-9320 elemental analyzer. Surface morphology and elemental composition of the sorbent were examined by scanning electron microscopy (SEM) coupled with energy-dispersive X-ray spectroscopy (EDX), using a Zeiss Evo HD15 system equipped with a tungsten electron source.
Ultrasound-assisted extraction procedures were performed in a Bandelin ultrasonic bath. Ultrapure water utilized throughout the experiments was supplied by a Thermo Scientific Smart2Pure Pro system. Quantitative analysis of metal ions was conducted by using a Varian 710-ES inductively coupled plasma optical emission spectrometer (ICP-OES). The emission wavelengths employed for the determination of Cu(II), Cd(II), and As(III) were 324.75, 228.80, and 193.70 nm, respectively.
Results and Discussion
Characterization of Si-Carb-Formazan
Structural characterization of the synthesized silica-based sorbent modified with a thiosemicarbazone (TSC) ligand was conducted by using FTIR spectroscopy and SEM-EDX analyses. As illustrated in Figure 3, the FTIR spectra provided clear evidence for the presence of both the silica support and the organic ligand. In the spectrum of unmodified silica (red), a broad absorption around 3436 cm–1 was attributed to O–H stretching vibrations of surface silanol groups, while a distinct peak near 1090 cm–1 indicated the asymmetric stretching of Si–O–Si bonds. Additional bands between 966 and 808 cm–1 were assigned to bending vibrations associated with Si–O and Si–OH functionalities.
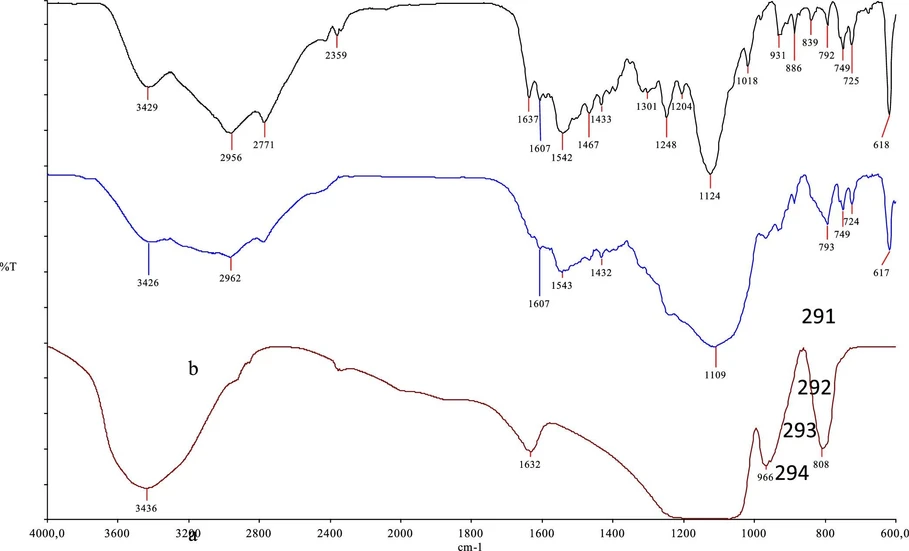 ACS Omega 2026, 11, 1, 1213–1235: Figure 3. Comparative FTIR spectra of the TSC ligand (black), functionalized silica material (blue), and unmodified silica (red) are shown from top to bottom, respectively.
ACS Omega 2026, 11, 1, 1213–1235: Figure 3. Comparative FTIR spectra of the TSC ligand (black), functionalized silica material (blue), and unmodified silica (red) are shown from top to bottom, respectively.
The pure TSC ligand spectrum (black) revealed a broad band at 3429 cm–1, corresponding to N–H stretching vibrations. A strong absorption at 1542 cm–1 confirmed the presence of the imine group (C═N), a key structural feature of the TSC. Moreover, the peaks at 2956 and 2771 cm–1 were consistent with aliphatic C–H stretching modes, and the range between 1248 and 1018 cm–1 showed characteristic C–N stretching vibrations.
In the spectrum of the functionalized silica material (blue), absorption features from both the silica matrix and the organic ligand were observed, indicating successful covalent grafting. The persistent presence of the imine C═N band at 1543 cm–1 demonstrated that the TSC retained its structural identity postimmobilization. The broad signal at 3426 cm–1 suggested overlapping of the O–H and N–H stretching vibrations from the silica and the ligand moieties. Furthermore, the intense band at 1109 cm–1 confirmed the structural integrity of the silica framework through Si–O–Si vibrations, and the peaks at 2962 and 2770 cm–1 supported the presence of aliphatic C–H groups associated with the ligand. Altogether, these spectroscopic observations conclusively verified the successful functionalization of the silica surface with the TSC ligand via chemical bonding interactions.
Morphological and elemental characteristics of the TSC-functionalized silica sorbent were evaluated both before and after metal ion adsorption using scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDX), and elemental mapping techniques. As depicted in Figure 4, the SEM micrograph obtained prior to metal binding revealed a relatively uniform and porous surface topology. Following the adsorption process, noticeable morphological changes were observed, including increased surface roughness and particle agglomeration, indicating the interaction between the sorbent and metal ions.
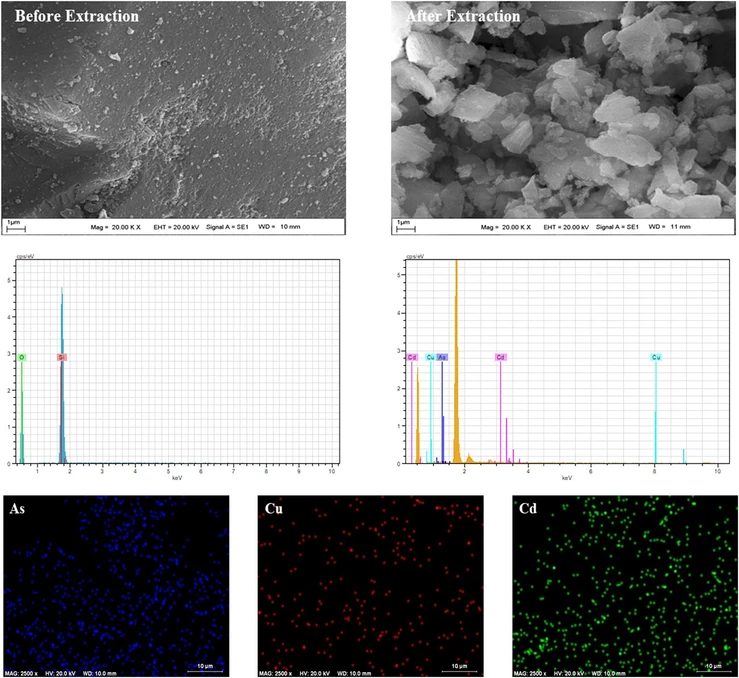 ACS Omega 2026, 11, 1, 1213–1235: Figure 4. SEM images and EDX spectra of the sorbent before and after metal sorption.
ACS Omega 2026, 11, 1, 1213–1235: Figure 4. SEM images and EDX spectra of the sorbent before and after metal sorption.
Furthermore, EDX spectral analysis of the pristine sorbent confirmed the presence of only silicon (Si) and oxygen (O), consistent with the silica matrix. However, after exposure to metal-containing samples, new peaks corresponding to arsenic (As), cadmium (Cd), and copper (Cu) appeared in the EDX spectrum, demonstrating that these metal ions were successfully captured by the sorbent. Complementary EDX mapping further confirmed that these metal ions were uniformly distributed across the surface of the material, providing visual evidence of effective and homogeneous adsorption. Collectively, these findings validate the selectivity and efficiency of the prepared sorbent for the adsorption of As(III), Cd(II), and Cu(II) ions from complex matrices.
C12H12BrN3S2: Calc. %C: 42.11; H, 3.53; N, 12.28; S, 18.73. Found: %C: 41.81; H, 3.42; N, 12.17; S, 18.75. FTIR (KBr pellet) ν/cm–1 3429 (N–H), 1542 (C═N), 1124 (N–N), 839 (C═S), 792 and 725 (thiophen and phenyl ring stretching). 1H NMR (400 MHz, DMSO-d6) data [δ 11.14 (s, 1H), 8.33 (s, 1H), 7.97–7.89 (m, 2H, ArH), 7.50–7.39 (m, 2H, ArH), 3.23 (s, 6H, N(CH3)2)] are correctly cited as Figure 5, whereas the 13C NMR (100 MHz, chloroform-d) data [δ: 173.89, 143.82, 137.17, 134.85, 131.50, 127.95, 125.69, 125.57, 122.63, 116.97, 40.18 (2 × C)] are cited as Figure 6.
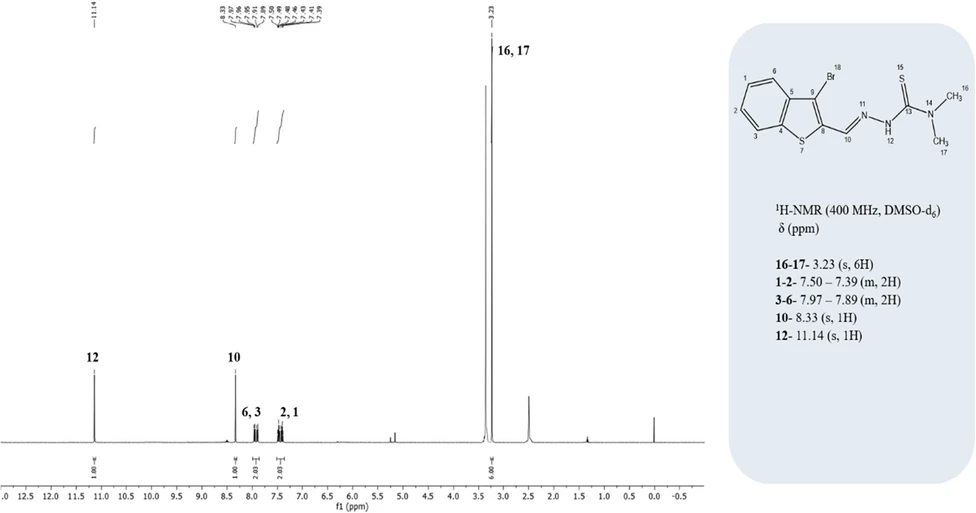 ACS Omega 2026, 11, 1, 1213–1235: Figure 5. 1H NMR spectrum of the synthesized thiosemicarbazone (TSC) ligand.
ACS Omega 2026, 11, 1, 1213–1235: Figure 5. 1H NMR spectrum of the synthesized thiosemicarbazone (TSC) ligand.
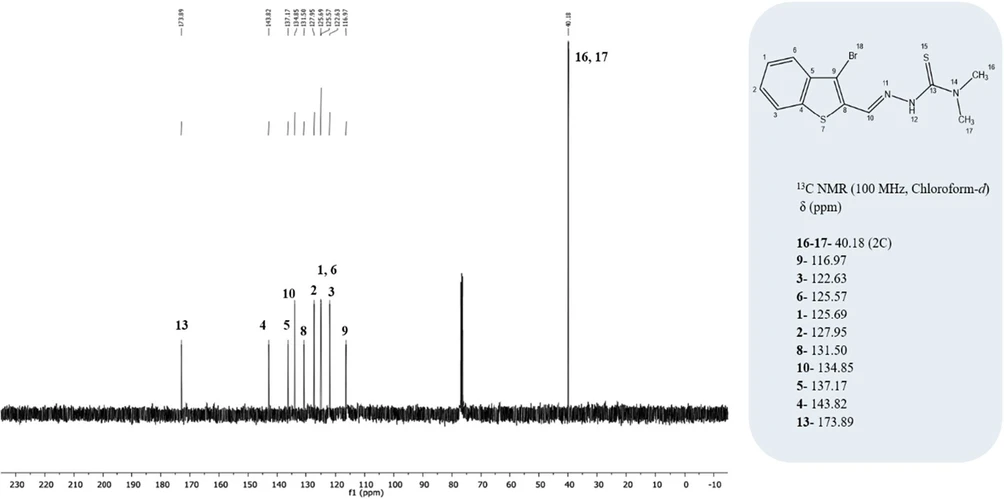 ACS Omega 2026, 11, 1, 1213–1235: Figure 6. 13C NMR spectrum of the synthesized thiosemicarbazone (TSC) ligand.
ACS Omega 2026, 11, 1, 1213–1235: Figure 6. 13C NMR spectrum of the synthesized thiosemicarbazone (TSC) ligand.
Conclusions
In this study, a novel thiosemicarbazone derivative was successfully synthesized and characterized using spectroscopic (FTIR, NMR) techniques. Its coordination behavior toward Cu(II), Cd(II), and As(III) ions was systematically investigated using the UA-DMSPE method, demonstrating high extraction efficiency and selectivity under optimized conditions. The method was effectively applied to real samples, including water, soil, and tomato matrices, highlighting its potential for environmental and food safety monitoring.
Computational studies provided theoretical insights into the stability and electronic features of the ligand with DFT calculations supporting and rationalizing the experimentally observed donor–acceptor interactions. Molecular docking analyses revealed moderate but specific binding affinities of the ligand toward biologically relevant proteins, suggesting potential pharmacological relevance in addition to its analytical applications.
Overall, the integration of synthesis, analytical validation, and computational modeling provides comprehensive insight into the multifunctional properties of this novel thiosemicarbazone derivative. The findings not only establish a reliable platform for trace metal monitoring in complex matrices but also open avenues for future investigations of thiosemicarbazones as dual-purpose ligands with both environmental and biomedical significance.