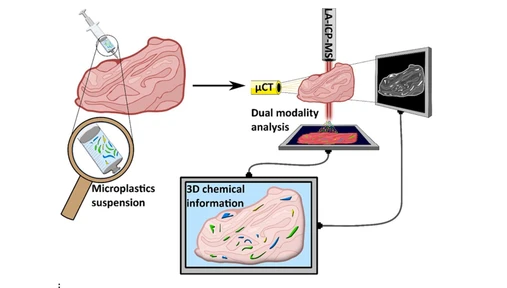
Investigating Plastic–Metal Interactions in Aquatic Environments Using Laser Ablation ICP–MS and Chemical Markers
ACS EST Water 2026, 6, 3, 1925–1935: Graphical abstract
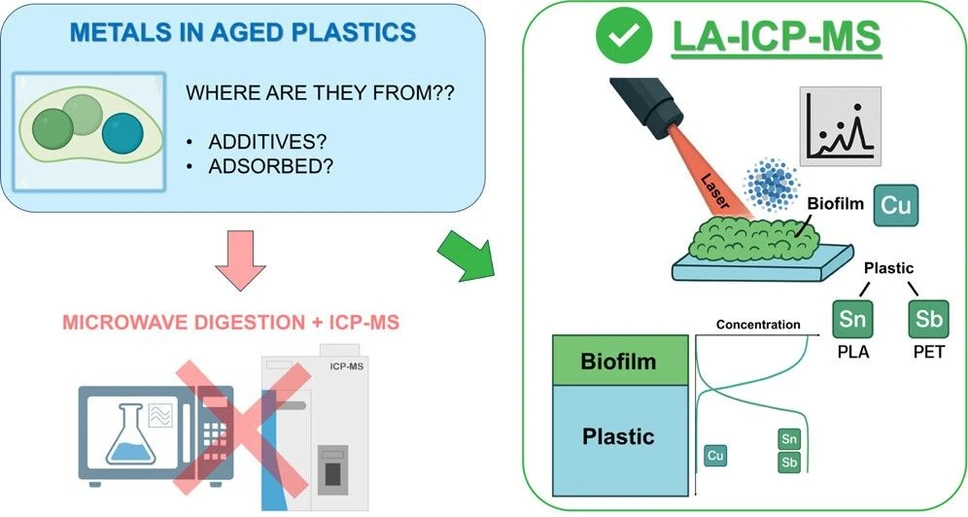
This study investigates interactions between plastics and metals in aquatic environments using laser ablation ICP-MS to map elemental distributions in biotically aged polymers. Copper was used as a marker of biofilm presence, while antimony and tin identified PET and PLA phases, respectively.
Results showed significant copper enrichment on aged plastic surfaces, confirming biofilm-mediated metal accumulation. Conventional bulk analysis underestimated this effect, highlighting the advantage of LA-ICP-MS for spatially resolved measurements. These findings emphasize the role of biofilms in metal transport and the importance of advanced techniques for environmental risk assessment.
The original article
Investigating Plastic–Metal Interactions in Aquatic Environments Using Laser Ablation ICP–MS and Chemical Markers
Davide Spanu, Ludovica Botta, Stefano Carnati, Tommaso Grande, Gabriela Kalčíková, Luca Nizzetto, Andrea Pozzi, Luka Šupraha, and Gilberto Binda*
ACS EST Water 2026, 6, 3, 1925–1935
https://doi.org/10.1021/acsestwater.5c01387
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
The interaction between plastic and (trace) elements in waters has attracted the interest of researchers: the possible implications, such as the alteration of natural biogeochemical cycling and the potential vector effect of toxic metals through the trophic chain, are worrying for ecosystem health. Their environmental relevance, however, is yet to be understood. (1−5)
Plastic has, in fact, been observed to actively accumulate metal ions from the environment in water bodies. The processes of plastic aging (i.e., the degradation and alteration of plastics by abiotic and biotic factors) play a pivotal role here. (2,6,7) In particular, biotic aging has been identified as a key determinant of the metal adsorption processes: (3) the colonization of plastic by a microbial community can completely alter the surface properties of plastic, providing different surface functional groups and altering surface hydrophobicity, increasing metal sorption from the environment. (8) In addition, several microorganisms are known to actively accumulate metal ions inside their cells as some of them are essential for their life. (9) As an example, Cu is an essential element (at environmentally relevant concentrations), and microorganisms can both internalize it in their cell tissues or immobilize it in the extracellular exudates, such as polysaccharides. (10−12)
In addition to adsorbing metals from the environment, plastics can contain several inorganic chemicals, such as functional additives, pigments, and impurities. (4,13,14) Other metals are instead added as a catalyst during the production of specific polymers: a known example is the use of Sb in polyethylene terephthalate (PET) to favor the polycondensation of this polymer. (4,15) These additives may leach out from the plastic matrix into the surrounding water, and the environmental aging of plastics appears to further influence these leaching processes. This further complicates the evaluation of potential risks related to plastic pollution. (16,17)
A crucial aspect of studying the interaction between plastics and metals is distinguishing between additives incorporated within the polymer matrix and metals accumulated from the environment. To address this knowledge gap, several studies have investigated environmental samples through bulk analysis (e.g., through acid digestion), as well as integrating specific leaching solutions. (18−20) Within this framework, laser-based techniques, such as laser ablation─inductively coupled plasma─mass spectrometry (LA–ICP–MS), are emerging as tools to resolve the microscale spatial distribution of both inorganic additives and trace elements sorbed from the environment. (21) Some recent applications showed the potential of LA–ICP–MS to track elemental content in pristine and aged plastic samples, including environmental samples. (22−25) While the interest in these techniques is increasing, there still are issues hampering a broader applicability of LA–ICP–MS to understand the interaction dynamics of metals and plastics, such as the complex and poorly known array of metal-containing additives present in plastics and the several processes affecting metal adsorption in natural environments (e.g., climate, pH, and particulate matter). (18,19,21,26)
In this study, we employed a LA–ICP–MS-based approach to trace the enrichment of trace metals on plastic materials aged in synthetic freshwater to allow biofilm growth. Laboratory-scale experiments simulating plastic biotic aging help in reducing ambiguity for this kind of experiment, while chemical markers can track the polymer matrix, allowing a detailed cross-sectional view of metal adsorption from the environment. Here, we used plastic-specific inorganic additives as valid markers to detect the polymer matrix (e.g., Sb in PET and Sn in PLA, present as catalyst residuals after polymer production (13,16,24,27)). These elements are in fact present in the plastic products at easily detectable concentrations (in the tens of mg/kg range). To observe the unique help of chemical markers in investigating plastic–metal interactions, we also investigated a marker-free plastic sample made of polypropylene (PP) in this experiment. We also monitored Cu concentration in plastic and in water as this trace element is known to enrich in biofilms. (28−30) By combining physicochemical characterization, total metal content analysis, and scanning electron microscopy, we investigated the biofilm thickness on plastic surfaces and their capacity to selectively accumulate elements from the surrounding water. Implementing these approaches, we aim to improve understanding of plastic–metal interaction dynamics and to demonstrate the potential and limitations of LA–ICP–MS for monitoring and profiling of metals at the plastic–biofilm interface. This investigation also sheds light on the key implications of biotic aging in affecting the properties of plastic pollutants.
2. Materials and Methods
2.3. Physicochemical Characterization of Plastic and Biofilms
After collection, all plastic samples were air-dried for 24 h and analyzed for their physicochemical properties to assess the changes induced by biotic aging. In addition, specific measures to analyze the physicochemical features of the biofilm community were investigated too.
To observe the changes in surface functional groups, we used Fourier transformed infrared spectroscopy in the attenuated total reflection (ATR-IR) mode (Nicolet iS 10, Thermo Scientific, Waltham, MA, United States of America). Thirty two scans were performed for every sample in the 4000–650 cm–1 spectral interval, with a resolution of 0.482 cm–1. A background spectrum was recorded prior to every measurement. Collected IR spectra were then normalized on the maximum absorbance peak using Origin 2024 pro software (OriginLab Corporation).
Scanning electron microscopy (SEM) images were collected to examine the surface micromorphology of plastic fragments with and without the presence of a biofilm as well as to understand the quantity of the ablated plastic material during the LA–ICP–MS analyses (see Section 2.6) using a Philips (Netherlands) field emission gun-scanning electron microscope (FEG-SEM), with a 20 keV beam under high vacuum conditions. Before SEM analysis, samples were made more conductive by covering them with a 5 nm thick gold layer using a Cressington (UK) 108 auto vacuum sputter coater.
Static water contact angles were also measured to assess the hydrophobicity of plastic samples using a 3D printed instrument. (34) Briefly, 3 μL of ultrapure water was deposited on the sample surface, and pictures were collected via a smartphone (Samsung Galaxy S21FE) camera. Then, contact angles were computed on the collected images using ImageJ software using the “drop_analysis” plugin. (35) Samples were collected in triplicate for each specimen.
2.4. Analysis of Total Metal Content in Plastic
The total metal content of plastic samples was assessed using a microwave-assisted acid digestion protocol set up by our research team (please also refer to this publication for detailed methods and quality protocols). (32) Plastic sample digestion was obtained through microwave-assisted heating in a closed system (ETHOS One, Milestone MLS, United States of America) equipped with polytetrafluoroethylene (PTFE) vessels. Briefly, approximately 100 mg was weighed and inserted into each vessel, and 4 mL of 65% v/v HNO3 and 1 mL of 95% v/v H2SO4 were added to the vessels. The materials were then digested by applying a temperature ramp, reaching 200 °C for 45 min. A further digestion was then performed by adding 0.1 mL of H2O2 to each vessel and leaving to react for 30 min at room temperature, after which another H2O2 addition of 0.1 mL was made. Solutions were then left at room temperature inside the vessels until cool.
2.5. Instrumental Analysis of Metals in Solution
Solutions obtained by the different aging batches and after acid digestions were then analyzed for their content of Sn, Sb, and Cu using an ICP–MS instrument. All these solutions were filtered (0.45 μm PTFE filter), acidified with HNO3 reaching a 2% v/v concentration, and spiked with two internal standards (Rh and Re, respectively). The solution was then finally analyzed with a Thermo Scientific ICAP Q (United States of America) ICP–MS, and metal(loid) quantification was obtained by external calibration. Details of the instrumental setup are listed in Table S2.
3. Results and Discussion
3.1. Impact of Biotic Aging on Plastic Physicochemical Properties
Biotic aging affected the physicochemical properties of all of the plastic samples. However, the extent of these alterations was strictly polymer-dependent. As a primary effect, the coating of the biofilm evidently influenced the surface morphology of plastic (Figure S1). SEM micrographs (Figure 1) show a thick layer of microorganisms homogeneously covering the plastic samples after the aging, which varied from about 5 μm up to about 50 μm depending on the plastic type. Such a difference in thickness may be induced by both plastic polymer hydrophobicity and micromorphology and the type of chemicals present as additives. In this study, PP and PLA showed comparable thickness, while PET showed a distinct lower thickness. This evidence is corroborated by both a significantly lower mass of the biofilm observed after Fenton digestion and a lower concentration of EPS in the PET-aged fragments in comparison with the samples made of other polymers (Figure S2). This may be induced by a lower hydrophobicity, potentially reducing microbial attachment, (28) the limited availability of carbon as a source for bacteria being a nondegradable polymer, (42) and the relatively high content of additives, such as Sb, which may potentially hamper a quick attachment of microorganisms on the polymeric substrates. (43) Observing the microbial community composition from SEM micrographs, several filamentous algae and coccoids were evident, as well as several residuals of diatom frustules (Figure S3b), as indicators of widespread colonization by microorganisms in these samples. (19,44) The pristine samples, on the other hand, show a smooth surface with only minor defects (Figure S3).
 ACS EST Water 2026, 6, 3, 1925–1935: Figure 1. SEM micrograph of PET (panels a, b), PLA (panels c, d), and PP plastics (panels e, f) after the biotic aging process. Figures in the left column (panels a, c, and e) show the frontal view of the thick biofilm layer formed on the samples, while figures on the right column (panels b, d and f) show the lateral view, highlighting the different biofilm thickness.
ACS EST Water 2026, 6, 3, 1925–1935: Figure 1. SEM micrograph of PET (panels a, b), PLA (panels c, d), and PP plastics (panels e, f) after the biotic aging process. Figures in the left column (panels a, c, and e) show the frontal view of the thick biofilm layer formed on the samples, while figures on the right column (panels b, d and f) show the lateral view, highlighting the different biofilm thickness.
Concerning the surface chemical properties, the IR spectra of all of the polymers showed the typical adsorption band of their composing polymers. For instance, the characteristic absorption bands located at 2960–2850 cm–1 and 1480–1300 cm–1 typical of C–H stretching of methyl and methylene groups for PP; (45) the absorption peak of strong intensity at 1714 cm–1 (stretching of C═O of the carboxylic acid group) or the esters stretch at 1240 cm–1 for PET; (46,47) and the band absorbing peaks at 1750 cm–1 and 1180 cm–1 related to stretching vibration of carbonyl (C═O) and ester (C–O) groups, as well as the peak at 1452 cm–1 assigned to bending of the –CH3 group for PLA (48,49) (Figure 2). The formation of the biofilm altered the prevalence of surface functional groups regardless of the polymer type. For example, amide I and amide II bands are evident at 1650 cm–1 and 1550 cm–1, respectively, in almost all the polymers after aging (Figure 2). These peaks also altered the representative peak at 1714 cm–1 of PET, which showed a marked shoulder after aging (Figure 2a). Similarly, changes in intensity and shape of the OH band at 3500 cm–1 were observed in all the polymers: the broad peak in the 3000–3500 cm–1 band results in a sharper profile after aging, as an index of the presence of free and bonded hydroxyl groups and structural hydroxyl groups (–COOH and─COH). (50) This is typical of the cell membranes and the extracellular polymeric substances associated with freshwater microorganisms, such as microalgae. (51) These changes are clearly attributable to the biofilm formation, especially if we consider the depth probed by the analysis through ATR-IR measurements, which probe the first few micrometers below the sample surface. As the thickness of the biofilm layer observed from the SEM measurements is in the range of about 5 to 45 μm (as observable in Figure 1), the IR spectra are predominantly affected by the presence of the functional groups of the biofilm layer. The analysis of the EPS content also confirmed such changes induced by the presence of these compounds in the biofilm matrix (Figure S2).
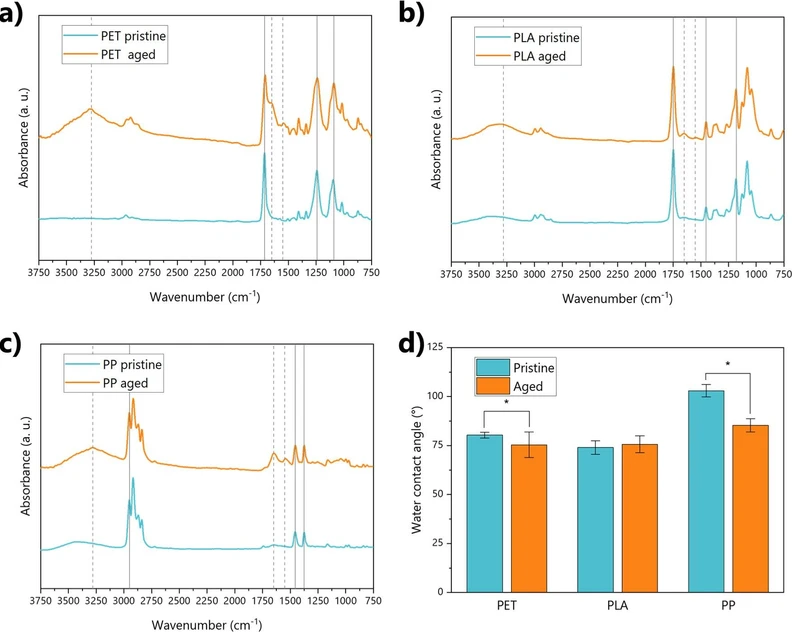 ACS EST Water 2026, 6, 3, 1925–1935: Figure 2. Physicochemical properties of pristine and aged plastics. Panels a, b, and c show the IR spectra of PET, PLA, and PP, respectively, before and after aging. Gray lines highlight the key functional groups of the polymers (if solid) and the functional groups affected by aging (if dashed). Panel d shows instead the values of water contact angles for all polymer samples. Significantly different values between pristine and aged plastics are highlighted by asterisks.
ACS EST Water 2026, 6, 3, 1925–1935: Figure 2. Physicochemical properties of pristine and aged plastics. Panels a, b, and c show the IR spectra of PET, PLA, and PP, respectively, before and after aging. Gray lines highlight the key functional groups of the polymers (if solid) and the functional groups affected by aging (if dashed). Panel d shows instead the values of water contact angles for all polymer samples. Significantly different values between pristine and aged plastics are highlighted by asterisks.
3.4. Advantages, Limits, and Future Developments for the Application of LA–ICP–MS
This study showcases the potential of LA–ICP–MS to complement common techniques to investigate the interactions of plastics and metals in aquatic ecosystems. This technique enabled us to highlight the marked enrichment in Cu on the surface of aged plastic, in comparison to pristine one, associated with the biofilm. A key advantage of LA–ICP–MS is its surface sensitivity combined with high analytical sensitivity. (21,23,41) In contrast, the traditional acid digestion protocol failed to quantify adsorbed species, as evidenced by Cu values consistently below the limit of quantification (Figure 3).
Depth-resolved capability of LA–ICP–MS enabled the distinction between biofilm-associated metal signals and bulk polymer contributions, providing also a quantitative estimation of biofilm thickness. By simultaneously monitoring biofilm markers (e.g., Cu) and polymer-specific additives (e.g., Sn in PLA, Sb in PET), the technique offers an array of complementary information to unambiguously discriminate between metal adsorption from the environment and additive diffusion within the polymer matrix: such a result is not achievable using conventional techniques. In other words, our results demonstrate that LA–ICP–MS can serve as a chemical marker-based approach particularly valuable for assessing surface versus bulk metal distributions, which is critical for understanding the environmental fate of specific elements in the context of widespread plastic contamination. This study represents a first step in this type of investigation, highlighting an approach with considerable potential that has so far been largely overlooked in the current literature. (24)
Nonetheless, there are some limitations to consider and future refinements to improve the applicability of this technique. The technique requires careful optimization of instrumental parameters (e.g., fluence, spot size, number of ablations) to avoid polymer melting or irreproducible ablation, and quantitative interpretation can be influenced by polymer-specific behavior. (54,55) Moreover, for polymers lacking systematically present metal additives (such as PP in this study), only biofilm markers (e.g., Cu) can be used to trace biofilm presence, limiting the ability to simultaneously monitor bulk polymer contributions. Additionally, for a more precise quantification of metals in the polymer matrix, the use of standard reference materials would be highly beneficial to calibrate ablation efficiency and account for matrix effects. (52,56) However, certified plastic materials with known trace element content are not available for many polymer types, and importantly, only their bulk content is known, with no information on the distribution of elements within the polymer matrix at the microscopic scale. (32) This information is crucial for LA–ICP–MS analysis. (54)
A further step to assess the reliability of this approach for investigating plastic–metal interactions and the key role of biofilms in mediating these processes would be to compare LA–ICP–MS with other high-resolution surface techniques capable of chemical mapping and depth profiling in these matrices, such as secondary ion mass spectrometry (SIMS) or synchrotron-based X-ray techniques. (57,58) Although these methods are substantially more costly and require more demanding sample handling and preparation, they offer higher spatial resolution and analytical sensitivity, complementing the insights obtained by LA–ICP–MS. In addition, while being less sensitive for specific metal markers, Raman microscopy may also be applied as a comparative approach to investigate biofilm-mediated environmental processes at plastic surfaces. (59)
4. Conclusions
Understanding the mechanisms by which plastics interact with metals is essential for properly evaluating their chemical risks, and LA–ICP–MS can offer a valuable tool to complement conventional characterization methods. In this study, we tested the feasibility of this approach under controlled laboratory conditions, using three different polymer types subjected to biotic aging and focusing on a set of metals to be used as markers of the biofilm (i.e., Cu) and of the polymer matrix (i.e., Sb and Sn). Our LA–ICP–MS results showed the marked accumulation of Cu from water on the biofilm layer covering aged plastics on all polymer types. This was confirmed by the marked decrease in Cu in the water phase, indicating its potential sorption. By contrast, conventional acid digestion revealed only limited amounts of Cu in the plastic samples, indicating the superior and unique performances of LA–ICP–MS in understanding plastic and metal interactions. The use of metallic chemical markers for plastic also proved useful in interpreting the patterns of metal enrichment associated with biofilm development, presenting a high potential for environmental applications. In summary, this study demonstrates the potential of LA–ICP–MS as a powerful technique for investigating plastic–metal interactions in controlled settings and highlights, for the first time, the combined use of polymer-specific markers and depth-resolved analyses to better understand metal enrichment processes driven by biofilms. This technique represents a promising surface-sensitive approach that can fill gaps left by conventional techniques. It provides unique insights into the spatial distribution of metals on and within plastic materials, enabling the study of biofilm-mediated sorption processes under environmentally relevant conditions.