Green Approaches for Preparation of Natural Deep Eutectic Solvents for Determination of As, Cd, and Pb in Plant Samples by ICP-MS
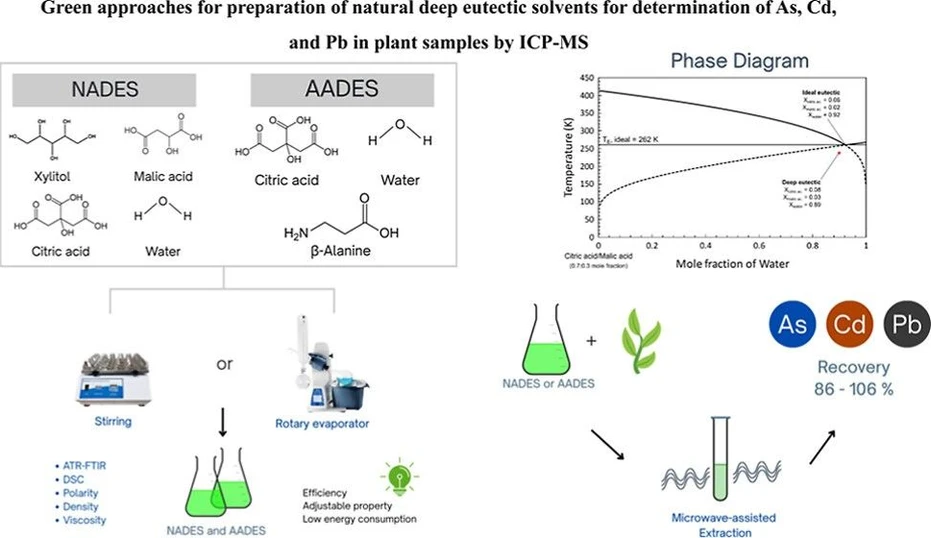
ACS Omega 2025, 10, 24, 26118–26128: Graphical abstract
Natural deep eutectic solvents (NADESs) and amino acid-based deep eutectic solvents (AADESs) are eco-friendly alternatives with low toxicity, low cost, and tunable physicochemical properties. In this work, two green preparation methods were developed using stirring without heating and a rotary evaporator under reduced pressure, with comparisons to the conventional heating approach.
The solvents, based on citric acid with β-alanine, xylitol, or malic acid, were characterized by FTIR, DSC, viscosity, and melting point measurements, confirming their eutectic nature. Both methods produced effective solvents for microwave-assisted extraction of plant materials, enabling recoveries of 87–106% for Cd, Pb, and As. EcoScale evaluation gave high green scores (99 for stirring, 98 for rotary evaporation), highlighting the efficiency and sustainability of these preparation methods.
The original article
Green Approaches for Preparation of Natural Deep Eutectic Solvents for Determination of As, Cd, and Pb in Plant Samples by ICP-MS
Sabrina S. Ferreira, Rafaela S. Lamarca, Leandro S. Silva, Thiago A. L. Burgo, Paulo C. F. Lima Gomes, Clarice D. B. Amaral, Jose O. Fernandes, Sara C. Cunha, Mario H. Gonzalez*
ACS Omega 2025, 10, 24, 26118–26128
https://doi.org/10.1021/acsomega.5c03345
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
Actions aimed at reducing the production, use, and exposure potential of harmful chemicals are essential for achieving sustainability goals. Unfortunately, conventional analytical methods often worsen environmental issues, because the production and use of toxic solvents contribute to the generation of hazardous waste during the analytical process. (1−3) For this reason, significant efforts are being made to develop new sustainable solvents to replace toxic ones, aiming at reducing the environmental impacts of analytical methodologies while maintaining the quality of analytical results.
A major challenge in green analytical chemistry (GAC) is the design and synthesis of sustainable solvents suitable for use with a wide variety of solutes. A sustainable solvent should comply with the principles of GAC, considering aspects including waste disposal, impacts on the environment and human health, and safety of use. (4)
Deep eutectic solvents (DESs) are currently considered a sustainable and economically attractive option, due to their low toxicity, biodegradability, and affordable synthesis. (5,6) A DES is a mixture of two or more organic compounds at a ratio close to the eutectic point, with a melting point lower than for each of the precursors alone. (7) These solvents are known as natural deep eutectic solvents (NADESs), when natural compounds such as sugars, organic acids, amines, and amino acids are used in the synthesis. (8) A NADES can be more strictly classified as an amino acid-based deep eutectic solvent (AADES), when an amino acid is used as one of the solvent precursors. (9,10)
The components of DES such as NADES and AADES interact primarily by means of hydrogen bonds, with one of the precursors serving as a hydrogen-bond donor (HBD) and the other as a hydrogen-bond acceptor (HBA). The intermolecular hydrogen bonding network allows for charge delocalization, which is described as the cause of the decreased melting points of DES. (8,11) Changing the molar ratio of the precursors, or adding water to the solvent, modifies the interactions between the components, resulting in changes in physicochemical properties such as density, viscosity, and melting temperature. (8,12)
Since the discovery of DES and NADES, many studies have explored different combinations of precursors, different syntheses, and optimization of parameters such as the component ratio and water volume. (13) The main methods described in the literature for the preparation of DES are stirring and heating, lyophilization, vacuum evaporation, microwave irradiation, and ultrasonication. The stirring and heating method involves mixing two separate components in the presence of water, followed by stirring and heating the solution on a magnetic stirrer, until a clear liquid is formed. (14) In the evaporation method, the components are solubilized in water and evaporated at a temperature of approximately 50 °C, using a rotary evaporator, followed by storage of the resulting liquid in a desiccator until it reaches constant weight. (8) In microwave-assisted NADES preparation, the precursors are irradiated in a closed system, at controlled power and temperature. (15,16) Solvent formation can also be induced by sound waves in ultrasound-assisted NADES preparation. (16)
Due to their adaptability, DES and NADES are gaining popularity in analytical chemistry, as well as in areas such as catalysis, electrochemistry, materials chemistry, and organic synthesis. (14,17) In the last ten years, there has been a significant amount of research on the use of DES and NADES as extraction solvents employed in the preparation of solid and liquid samples. (13,18) Most of the studies have focused on the extraction of organic substances such as pesticides, drugs, bioactive substances, and phenolics. (19,20) The use of DES and NADES for the extraction of inorganic analytes has also been investigated in several studies. (21−23) Liquid-phase microextraction (LPME), ultrasound-assisted microextraction (UAME), and microwave-assisted extraction (MAE) are frequently mentioned in the literature, among many other sample preparation techniques using DES as extraction solvents. (10,24,25)
The number of studies exploring the applicability of DES has been increasing annually, but the influence of the physicochemical properties of DES has not yet been fully elucidated. (26) Therefore, the aim of this study was to investigate the physicochemical properties of NADES and AADES based on xylitol, β-alanine, and citric acid, prepared using two different routes. Investigation was made of the physicochemical properties of these solvents, including freezing point, density, viscosity, and polarity, and the solvents were compared with others prepared using the widely employed stirring with a heating method. The sustainability and green characteristics of the developed preparation methods of the solvents were evaluated using the EcoScale metric. (27) In addition, the extractive capacities of the solvents prepared by the proposed methods were evaluated in the analysis of plant tissue material using microwave-assisted extraction (MAE), followed by determination of toxic elements by ICP-MS.
2. Material and Methods
2.5. Characterization of the NADES and AADES
Infrared spectra of the NADES and AADES were obtained using a Fourier transform infrared spectrometer (Spectrum Two, PerkinElmer, USA) operated in attenuated total reflectance (ATR) mode, in the range 4000 to 400 cm–1, with a resolution of 4 cm–1 and an accumulation of 64 scans.
Density measurements of the NADES and AADES were performed (in triplicate) using a pycnometer calibrated with water, at 24 °C, and an analytical balance with a precision of ±0.0001 g (model AG200, Gehaka, São Paulo, Brazil). Viscosity measurements of the solvents were made (in triplicate) using a Cannon-Fenske viscometer calibrated with water, at 24 °C.
The melting points of the solvents were determined by differential scanning calorimetry (DSC), using a TA2010 instrument controlled by a TA5000 module (TA Instruments, USA). A 10 mg portion of the sample was placed on an aluminum support and heated from −100 to 400 °C, at 10 °C min–1, under a flow of N2 (100 mL min–1).
2.6. Sample Preparation and Analysis by ICP-MS
2.6.1. Microwave-Assisted Extraction (MAE) with NADES and AADES
The solvents prepared by stirring and rotary evaporator were used in a microwave-assisted extraction method to prepare a sample of a forage grass reference material (Brachiaria brizantha cv. Marandu, E1001a) supplied by the Brazilian Agricultural Research Corporation (Embrapa, São Paulo, Brazil), following the procedures described by Santana et al. and Guimarães et al. (21,28) For the Xyl-CA and MA-CA NADES, 90 mg of the sample and 9 mL of the solvent were used, with the following heating program: (I) 2 min ramp to 100 °C and (II) maintaining at 100 °C for 18 min (Santana, 2020). For the Ala-CA AADES, 200 mg of the sample and 5 mL of the solvent were used, with the heating program: (I) 2 min ramp to 100 °C and (II) maintaining at 100 °C for 40 min (Guimarães et al., 2022). After extraction, the suspensions were filtered, and the supernatants were appropriately diluted in ultrapure deionized water, prior to analysis by ICP-MS. The procedure was performed in triplicate.
2.6.2. ICP-MS Analysis
The extracts were analyzed by using a NexION 300X ICP-MS system (PerkinElmer, USA) equipped with a concentric nebulizer, a cyclonic nebulization chamber, and a quartz torch with a quartz injector tube (2.0 mm). The instrumental parameters were adjusted according to the manufacturer’s recommendations. The isotopes 75As+, 111Cd+, and 208Pb+ were analyzed in standard mode. The ICP-MS operating conditions are listed in Table 2. Calibration curves were prepared using dilutions of a 1000 mg L–1 stock standard solution of all of the analytes (Sigma-Aldrich, USA). For matrix matching, the calibration curves for analysis of the extracts were prepared using the standards in a NADES medium (1% v v–1).
3. Results and Discussion
In this study, two preparation methods for NADES and AADES were developed: stirring and a rotary evaporator. These methods were compared with preparation by stirring and heating, which is a method commonly reported in the literature.
3.1. Eutectic Characteristics and Physicochemical Properties
FTIR analysis was employed to investigate the intermolecular interactions and functional groups of the eutectic mixtures. The FTIR spectra of the precursors and NADES and AADES prepared by the three different methods (stirring, stirring with heating, and under reduced pressure in a rotary evaporator) are presented in Figure 1.
 ACS Omega 2025, 10, 24, 26118–26128: Figure 1. ATR-FTIR spectra of the initial reagents and the NADES and AADES prepared using the different methods for (a) Xyl-CA, (b) MA-CA, and (c) Ala-CA.
ACS Omega 2025, 10, 24, 26118–26128: Figure 1. ATR-FTIR spectra of the initial reagents and the NADES and AADES prepared using the different methods for (a) Xyl-CA, (b) MA-CA, and (c) Ala-CA.
The limit between the formation of the eutectic mixture and only solubilization of the components was investigated by applying the stirring procedure for a time until complete homogenization of the precursors, which was 20 min. The FTIR spectrum of the mixture obtained (Figure 2) showed no shift of the C═O bond peak at 1696 cm–1 to longer wavelengths, in addition to the peak having a lower intensity. This suggested that there was no significant increase in the electron density of carbonyl oxygen for the formation of hydrogen bonds between the precursors, which was a characteristic expected for effective formation of the solvent, under the conditions employed.
 ACS Omega 2025, 10, 24, 26118–26128: Figure 2. Comparison of the ATR-FTIR spectrum for the MA-CA NADES prepared by the stirring and heating method and the spectrum for the same components only solubilized under stirring.
ACS Omega 2025, 10, 24, 26118–26128: Figure 2. Comparison of the ATR-FTIR spectrum for the MA-CA NADES prepared by the stirring and heating method and the spectrum for the same components only solubilized under stirring.
It has been suggested that the formation of a deep eutectic solvent cannot be confirmed only by evidence of hydrogen-bond formation and by the eutectic point of the mixture being lower than the melting points of the individual components. It is necessary for the solvent to present a eutectic point temperature below the eutectic temperature of the ideal liquid mixture, with significant negative deviations from ideality, for it to be considered a deep eutectic solvent. (26) Therefore, analyses were performed to identify the melting points, calculate the ideal eutectic points, and obtain the phase diagrams of the proposed mixtures.
The NADES and AADES were submitted to DSC measurements at low temperature to identify the melting points of the mixtures obtained by the different preparation methods. Figure 3 shows the DSC curves for the Ala-CA AADES. The Xyl-CA and MA-CA NADES prepared by using a rotary evaporator under reduced pressure showed instability in solution, with phase separation occurring after 15 days of storage. This observed phase separation suggested disruption of the intermolecular interactions of the solvent, which precluded accurate melting point determination for these systems. The melting points obtained for NADES and AADES prepared using the three different methods are presented in Table 3.
 ACS Omega 2025, 10, 24, 26118–26128: Figure 3. DSC curves for the Ala-CA AADES obtained using the different methods: (a) stirring, (b) stirring/heating, and (c) rotary evaporator under reduced pressure.
ACS Omega 2025, 10, 24, 26118–26128: Figure 3. DSC curves for the Ala-CA AADES obtained using the different methods: (a) stirring, (b) stirring/heating, and (c) rotary evaporator under reduced pressure.
4. Conclusions
Green and sustainable methods were evaluated for the preparation of NADES and AADES, with the ability to modulate the physicochemical properties of the solvents produced. Stirring and rotary evaporator under reduced pressure preparation methods were employed to obtain three different NADESs and AADESs, based on combinations of citric acid, xylitol/malic acid/β-alanine, and water, and they were compared with the method using stirring with heating. A careful characterization of the solvents was made once the formation of the solvents using the stirring and rotary evaporator methods had been confirmed by FTIR and DSC analyses, together with the construction of phase diagrams.
When these solvents were employed in a method for microwave-assisted extraction of a plant material, they showed good capacity to extract Cd and Pb, using NADES prepared by stirring, and As and Pb, using AADES prepared by the stirring and rotary evaporator methods. The stirring and rotary evaporator methods for preparation of NADES and AADES have advantages in terms of efficiency and ease of use when compared to the conventional method of stirring with heating. Elucidation of the mechanisms of interaction between the components for the formation of NADES, the mechanisms of interaction between NADES and elemental analytes, and the factors involved in NADES–sample interactions are steps to be addressed in future studies necessary for the effective implementation of these solvents in sample preparation.