Recent development of CE-ICP-MS in biospeciation research and analysis: From anticancer drugs to nanoparticles and beyond
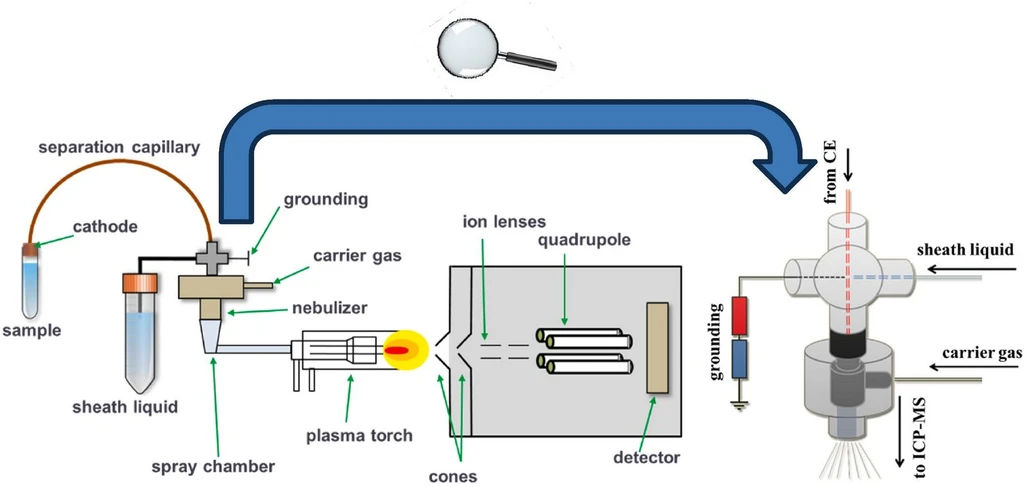
TrAC Trends in Analytical Chemistry, Volume 180, 2024, 117967: Fig. 1. Common scheme of CE-ICP-MS hyphenation workflow, with the detailed construction of the cross-connection.
The goal of this review is to critically assess the current and emerging capabilities of capillary electrophoresis coupled with inductively coupled plasma mass spectrometry (CE-ICP-MS) in biospeciation analysis. It focuses particularly on applications involving metal-based drugs and nanoparticles, where CE-ICP-MS offers simultaneous detection of chemical species in complex biological samples.
The review explores advantageous separation and detection strategies, highlights recent real-world applications, and discusses trends such as the growing use of tandem and single-particle ICP-MS. It also considers the potential for CE-ICP-MS to evolve into a robust, versatile method suitable for routine use in biospeciation research.
The original article
Recent development of CE-ICP-MS in biospeciation research and analysis: From anticancer drugs to nanoparticles and beyond
Magdalena Matczuk, Lena Ruzik, Andrei R. Timerbaev
TrAC Trends in Analytical Chemistry, Volume 180, November 2024, 117967
https://doi.org/10.1016/j.trac.2024.117967
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
The value of capillary electrophoresis (CE) in combination with ICP-MS as a powerful tool of speciation analysis was unlocked soon after its practical implementation in the late 1990s [1,2]. However, only following the commercialization of interface systems, was this hyphenated method recognized as practical for species-selective measurements in real-world biological samples [3]. Over the next decade, advances in ICP-MS used as a detector for CE ensured reliable speciation information from various biological materials, particularly in cases where extremely high sensitivity is not the issue [4]. A representative example of biospeciation challenges tackled by CE-ICP-MS is specific distributions of metal species produced by metal-based therapeutic drugs or diagnostic agents undergoing transformations in the body [5,6]. There are several advantages of CE-ICP-MS in this and the other research domains commonly referred to in comparison with HPLC-ICP-MS as a premier technique in the field [[7], [8], [9]]. They include (i) separation conditions both species-friendly, compatible with the physiological environment of analytes, and match well the ICP operation; (ii) good tolerance to biological matrices, typically requiring no pretreatment other than simple dilution; (iii) greater resolving power, in particular, regarding differently charged species; (iv) superior possibilities for analyzing microsamples; (v) simplicity in separation hardware (including its easy renewal), etc. Such benefits blended with a highly intensive, element-specific, and interference-free response of ICP-MS make the method tailor-made to elucidate the essentiality, toxicity, biological fate, and functions of trace elements and their compounds. It should not go without mentioning that apart from HPLC and CE, other separation techniques, such as gel electrophoresis or field-flow fractionation, find application in biospeciation research and analysis due to hyphenation with ICP-MS. However, in this field-specific review, we prefer to avoid ‘apples-and-oranges' comparisons but refer the interested reader to relevant review work [[10], [11], [12]].
The movement of CE-ICP-MS forward with solving practical speciation problems continued over the second decade of the 21st century. The progress accentuates the necessity to summarize and critically discuss the recently accumulated data. Such a review also appears timely because, since the last comprehensive coverage back in 2013 [4], there have been only a few literature examinations on CE-ICP-MS, in which speciation studies received a share of attention [[13], [14], [15], [16]], the most recent being published in 2021. Therefore, our primary goal here is to provide coverage of the latest literature for the period between 2019 and early 2024, with due account of examples indicating the speciation scope of CE-ICP-MS procedures, appropriate handling of any technical issues and limitations, and adequate method performance characteristics and acceptance limits. In addition, several earlier reports are brought into focus, proving the most advanced experimental approaches for speciation studies performed in highly controlled biological environments. Separation strategies and instrumental ICP-MS amendments required to meet best element-speciation practice will also be given special consideration. On the other hand, the technical setup and methodological requirements of CE-ICP-MS are not addressed here, as these issues were detailed in our previous review [4]. For the benefit of those who are not experts in the field, we deem it supportive to include a scheme showing the workflow of CE-ICP MS presented in Fig. 1. To highlight recent achievements and trends in this still expanding area, the CE-ICP-MS-based speciation assays are assembled and conferred below in different analyte categories, viz., metal-based drugs, metal-based nanomaterials, and metal ions playing various roles in human health.
 TrAC Trends in Analytical Chemistry, Volume 180, 2024, 117967: Fig. 1. Common scheme of CE-ICP-MS hyphenation workflow, with the detailed construction of the cross-connection.
TrAC Trends in Analytical Chemistry, Volume 180, 2024, 117967: Fig. 1. Common scheme of CE-ICP-MS hyphenation workflow, with the detailed construction of the cross-connection.
3. Speciation of metal nanoparticles
As ‘a new trick for the old dog', the most rapidly emerging application area of CE-ICP-MS is nanomedicine, one of whose pillars, metal-based nanoparticles, have received a great deal of attention from analytical chemists [[40], [41], [42], [43], [44]]. Whatever their intended biomedical application, i.e., disease prevention, diagnosis, therapies, etc., nanomaterials need to be well characterized regarding chemical transformations in the body to ensure their therapeutic efficacy and biosafety. Table 1 summarizes information on various nano–bio species originating from different (mostly metallic) nanoparticles in a biological fluid environment, together with separation and detection conditions, as well as some analytical figures of merit (where available). Note that no consideration is given here to the physicochemical characterization of nanoparticles, regarding the average size, size distribution, elemental composition, etc., or to their one-sort-but-different-size separations [55]. (Only single-size particles are supposed to be subject to administration.) Likewise, we do not consider the studies in which CE-ICP-MS is used for the quality assessment of newly synthesized nanoparticles [56,57].
4. Speciation of metals, metalloids and non-metals
Element speciation analysis can be read as an ‘old trick' of CE-ICP-MS [69]. While remaining the next best choice after HPLC-ICP-MS, a benchmark technique for the field [[70], [71], [72], [73]], the current CE-ICP-MS methodology mitigates the procedural problems encountered in speciation measurements of low-molecular-weight species from various materials of biotic origin (Table 2). It should be noted that the analytes shown in the table are those amenable to quantification. Others may have been attempted but fell below the limit of quantification of the developed procedure (or their determination was beyond the proposed task).
Several appealing contributions dealing with the speciation of transition metals came from Prof. Michalke's laboratory. The redox forms of iron were reliably quantified in the clinical samples of cerebrospinal liquid after the method's careful optimization and validation [77,78]. Used for this purpose were neuroblastoma cell lysates run with an acidic capillary electrolyte, following combined sample conductivity/pH-stacking, and recorded with ICP-MS, operating in the dynamic reaction cell mode (to alleviate a56ArO + spectral interference). The accuracy was verified by independent measurements with the validated HPLC-ICP-MS method. However, when turning to high-salt cerebrospinal liquid, the shift in migration times made mandatory standard additions to identify the iron species (Fig. 5). iron along with another redox active metal, manganese, and possibly neurotoxic zinc, were chosen for chronic and acute treatment of a model organism (Caenorhabditis elegans) to study the cellular response to imbalanced metal homeostasis linked to neurodegenerative diseases [80]. Using a similar CE-ICP-MS system, the signals of inorganic metal species were differentiated, and the in vivo alterations in the metallome were monitored. In contrast to iron, most of the zinc and manganese in the metal-treated worms were found loosely bound. The CE-ICP-MS method was also shown feasible to quantify the low doses of Cu, Mn, and Zn glycinates supplemented with animal feed (a mixture of corn flour and soybean meal) and to measure the interconversion between complexes and free metal ions [74].
![TrAC Trends in Analytical Chemistry, Volume 180, 2024, 117967: Fig. 5. Iron speciation in a cerebrospinal liquid sample (blue line). Standard additions: Fe(II) (red line) or Fe(III) (brown line). Reprinted with permission from Ref. [77]. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)](https://icpms.labrulez.com/labrulez-bucket-strapi-h3hsga3/Tr_AC_Trends_in_Analytical_Chemistry_Volume_180_2024_117967_Fig_5_Iron_speciation_in_a_cerebrospinal_liquid_sample_blue_line_be6706af25_l.webp) TrAC Trends in Analytical Chemistry, Volume 180, 2024, 117967: Fig. 5. Iron speciation in a cerebrospinal liquid sample (blue line). Standard additions: Fe(II) (red line) or Fe(III) (brown line). Reprinted with permission from Ref. [77]. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
TrAC Trends in Analytical Chemistry, Volume 180, 2024, 117967: Fig. 5. Iron speciation in a cerebrospinal liquid sample (blue line). Standard additions: Fe(II) (red line) or Fe(III) (brown line). Reprinted with permission from Ref. [77]. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
5. Conclusions
The CE's ‘love affair' with ICP-MS is long and storied, and the use of CE-ICP-MS in biospeciation research keeps its pace. However, as is true for every mature analytical technique, the method's progress largely depends on how significant the application base is. The past few years have witnessed a growth in the importance of the speciation of nanoparticles that are combined with metal-based drugs in some biomedical applications. From our examination of recent literature, the reader will gain an appreciation that this area tends to become a ‘killer' application of CE-ICP-MS, capturing the attention of a growing number of method practitioners – and this is one of the main messages of this review.
In the authors' opinion, there is not much room to optimize CE-ICP-MS instrumentation further, including the interface systems. Therefore, to bridge the present gaps in understanding the biological fate and effects of metal species or to define the biomedical potential of a particular nanomaterial, we should rather rely on refining the existing approaches than on promoting instrumental developments. Basic validation and quality assurance mechanisms can quickly meet good element-speciation practices, especially due to a lack of certified reference materials. These would make reliable and robust speciation CE-ICP-MS assays viable if executed commonly. Another limitation arises from more challenging biochemical tasks to resolve using the speciation data acquired, such as getting insight into the mechanism of neurodegenerative diseases. In this context, it is essential to emphasize that the major research endeavors stem from the groups of analytical chemists working in academic laboratories, who are not obligatory biological or medicinal chemistry experts. Therefore, if the proposed end-users, possessing such expertise, were to become more involved, then the results of the speciation measurements could be understood and appropriately implemented. The bottom line is that this review will help not only cover the current literature in the field but also inspire future research and development efforts.