Integrated laser ablation and computed tomography: detecting the chemical footprint of microplastics and reconstructing 3D biological tissues
Talanta 308, 2026, 129794: Graphical abstract
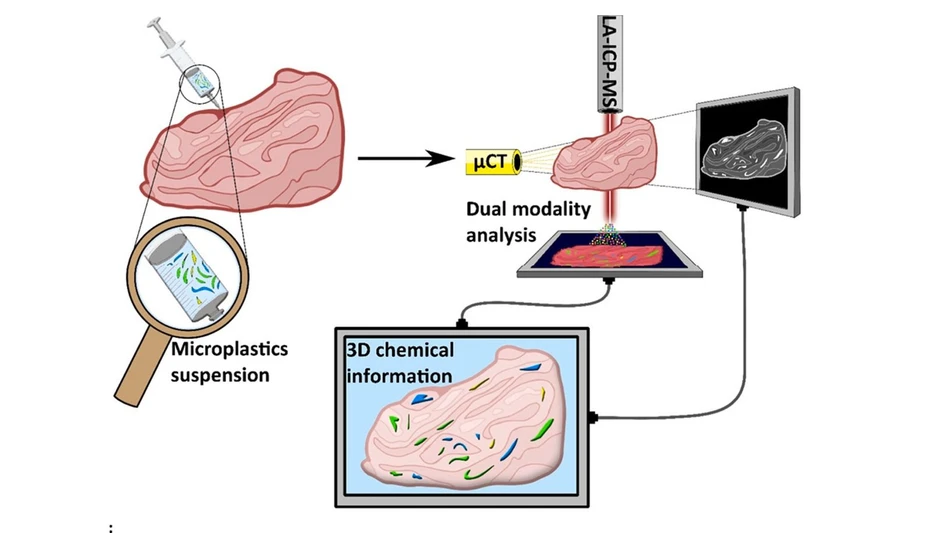
This study introduces a multimodal workflow combining micro-computed tomography (μCT) and laser ablation ICP-MS to detect and characterize microplastics in biological tissues. While μCT provides noninvasive 3D structural imaging, LA-ICP-MS enables chemical identification using metal markers such as antimony and cobalt in PET microplastics.
The integrated approach allows precise localization, segmentation, and size distribution analysis of microplastics within tissue. Validation against reference methods confirms improved detection capabilities, demonstrating the potential of this dual-modality strategy for comprehensive in situ analysis of microplastics in complex biological matrices.
The original article
Integrated laser ablation and computed tomography: detecting the chemical footprint of microplastics and reconstructing 3D biological tissues
Jan Biskupič, Peter Scheer, Jana Hložková, Ahmet Davut Aksu, Lucie Šudomová, Viktória Parobková, Petra Procházková, Tomáš Zikmund, Jozef Kaiser, Gabriela Kalčíková, and Michaela Kuchynka
Talanta 308, 2026, 129794
https://doi.org/10.1016/j.talanta.2026.129794
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
Microplastics (MPs) have become a defining environmental and biomedical concern of the 21st century. MPs, they are generated by degrading larger plastics or directly introduced through industrial and consumer products, and are now detected in every environmental compartment, from oceans and soils to food chains and drinking water [1]. Their presence has also been confirmed in organisms, including humans, raising urgent questions about their biodistribution, persistence, and toxicological effects [1]. While evidence suggests that MPs may accumulate in tissues and interact with biological systems [2], their transport mechanisms and long-term health implications remain unresolved. A major barrier to progress is the lack of robust, reproducible analytical workflows capable of simultaneously resolving MP structural localization and chemical composition in complex biological matrices.
Conventionally, biotic samples are digested using acidic, alkaline, oxidative, or enzymatic treatments, allowing the remaining particles to be inspected [3]. Initially, microscopy-based (e.g., optical microscopy and electron microscopy) methods were used [4]. However, it was not possible to distinguish MPs from other natural particulates; thus, chemical detection methods were incorporated. Spectroscopic methods such as Fourier-transform infrared (FTIR) or Raman spectroscopy are widely applied to obtain information on the size and polymer composition of MPs recovered from biotic and environmental samples [5,6]. Another suitable approach is the use of thermal-based techniques such as pyrolysis–gas chromatography–mass spectrometry (py-GC-MS), which does not require digesting samples, allowing small pieces of tissues to be analyzed directly. Such analysis provides quantitative information on the total mass of MPs and the relative abundance of different polymer types [7]; however, it does not provide information on MP size, which is essential for understanding the mechanism underlying their effects. Recently, several different combinations of analytical approaches have been proposed to better evaluate the distribution of MPs in biota.
Beyond standard techniques, recent advancements have highlighted the potential of laser-based or tomography techniques for characterizing and determining MP distribution. Laser-induced breakdown spectroscopy (LIBS) has been shown to provide elemental information on MPs [8]. Similarly, laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS) enables spatially resolved elemental mapping, offering enhanced sensitivity for detecting polymer-associated marker elements [9]. These methods are suitable for analyzing a variety of derivatized or native sample types (e.g., digested tissue or epoxy resin mount). Their limitation lies in the lack of a third dimension of spatial distribution. On the other hand, micro-computed tomography (μCT) has been shown to be a suitable technique for characterizing and assessing MP volume and distribution in biological tissues or even the whole organ systems of an animal [10]. However, volumetric data lacks important chemical information for differentiating MPs from other foreign objects or different polymer types.
In this context, the aim of this work is to establish a methodological framework that integrates two distinct analytical techniques with different sample preparation requirements. Initially, it is important to assess whether a single model biological sample can be sequentially analyzed using both methods. Therefore, μCT will be employed to obtain spatial information and identify potential MP locations within the spiked tissue. Subsequently, the same samples will be sectioned and analyzed via LA-ICP-MS to confirm the chemical composition of the detected particles, thereby verifying that they are indeed MPs. By correlating the spatial data from μCT with the compositional information from LA-ICP-MS, this study introduces, for the first time, a methodology that enables us to visualize the three-dimensional distribution of MPs in model biological tissues via chemically verified identification.
2. Materials and methods
2.1. Experimental workflow
The experiment was designed to visualize MPs within model tissue using two analytical approaches: μCT and LA-ICP-MS (Fig. 1). First, metal markers were quantified based on PET dilution and droplet formation, followed by element content analysis using LA-ICP-MS (Subchapter 2.2). The tissue sample was spiked with milled PET particles (Subchapter 2.3) and subsequently stained in an ethanolic iodine solution (Subchapter 2.4) to enhance the contrast in the soft tissue. μCT analysis was then performed to identify regions of interest (ROIs) with a high probability of MP accumulation (Subchapter 2.5). The tissue was embedded to the paraffin blocks and sectioned at the identified ROIs based on these findings, and thin sections were deparaffinized for LA-ICP-MS analysis (Subchapter 2.6). The elemental distributions obtained via LA-ICP-MS served as reference data for retrospective confirmation of MPs in the μCT datasets, enabling subsequent volumetric segmentation and three-dimensional modeling of MPs within the tissue, including statistical evaluation of particle size distribution (Subchapters 2.7 and 2.8).
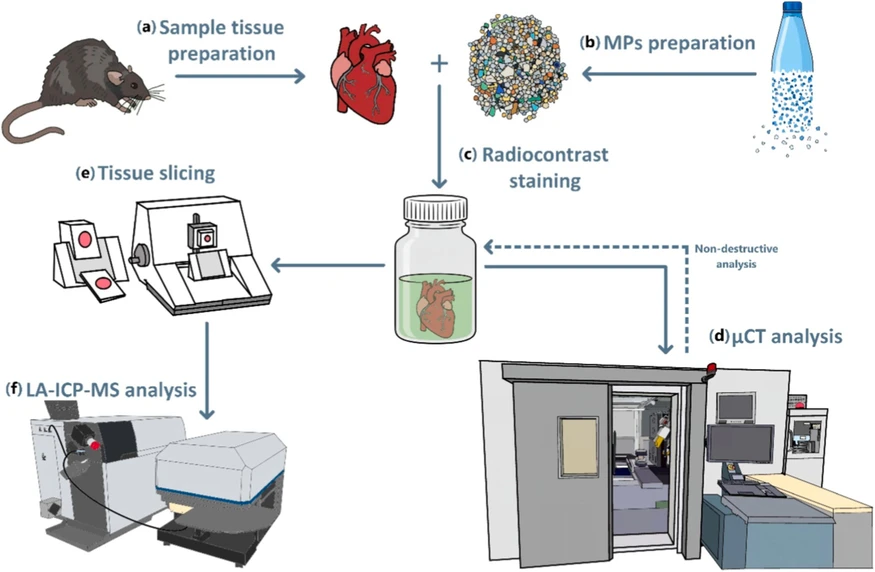 Talanta 308, 2026, 129794: Fig. 1. Workflow of the experimental procedure. (a) Tissue was collected from the model animal (rat). (b) The collected plastics were milled into MPs and then separated through a sieve with a defined mesh-size diameter. (c) The tissue was then spiked with MPs and immersed in a radiocontrast solution. (d) It was analyzed on a μCT instrument. (e) Then, it was embedded in paraffin and sliced using a microtome, and the sliced tissue was deparaffinized in xylene. (f) The deparaffinized tissue slide was analyzed on an LA-ICP-MS instrument.
Talanta 308, 2026, 129794: Fig. 1. Workflow of the experimental procedure. (a) Tissue was collected from the model animal (rat). (b) The collected plastics were milled into MPs and then separated through a sieve with a defined mesh-size diameter. (c) The tissue was then spiked with MPs and immersed in a radiocontrast solution. (d) It was analyzed on a μCT instrument. (e) Then, it was embedded in paraffin and sliced using a microtome, and the sliced tissue was deparaffinized in xylene. (f) The deparaffinized tissue slide was analyzed on an LA-ICP-MS instrument.
2.7. LA-ICP-MS analysis
The elemental distributions of cobalt (Co), antimony (Sb), and iodine (I) were determined using an LA-ICP-MS system. Co and Sb were selected based on the results of the first experiment (Subchapter 3.1), where I indicates the tissue. The setup consisted of a commercial LSX-213G2+ laser ablation unit (Teledyne Photon Machines, USA) equipped with a Q-switched Nd:YAG laser operating at a wavelength of 213 nm. The ablated material was transported in helium carrier gas into a single quadrupole ICP-MS 7900 spectrometer (Agilent, Japan). Sample imaging was performed under optimized ablation conditions, with integration times set for the isotopes 59Co, 121Sb, and 127I, and instrumental parameters (Table 1) were optimized following the procedures described by Vaculovic et al., 2015 [15]. The influence of variable ablation rates across different sample zones was considered according to the study by Anyz et al., 2017 [16], where the laser energy was optimized for full-slide thickness ablation across the sample zones. This optimization step was conducted on a small area of the slide before the analysis. Laser ablation synchronization with mass spectrometer dwell time was optimized according to the study by Lear et al., 2012 [17] to reduce the analysis time and minimize image aliasing. Raw data from both the laser ablation system and the mass spectrometer were evaluated using Iolite 4 (v4.10.4, Elemental Scientific Lasers) software, including baseline subtraction, element distribution maps, and multielement overlay maps. Following integration of element maps into microscopic mosaic and other graphical results, propagation were performed using Krita (v5.2.6) graphics software.
3. Results and discussion
3.3. Tissue slice evaluation via element-specific mass spectrometry imaging
Element-specific mass spectrometry imaging was utilized to determine the spatial distribution and chemical composition of the identified foreign objects (Fig. 3), including signal-to-noise ratio (SNR) values. The 127I signal of the stained tissue for μCT served as a high-intensity morphological reference (approx. 500 000 CPS; SNR = 2.5), clearly showing the biological tissue structure and boundaries. The first ROI (Fig. 3a) was chosen to cover the entire thickness of the tissue, allowing possible MPs that are deep inside the tissue to be determined. However, their presence was not confirmed, as no 121Sb or 59Co signals were found directly inside the tissue. In the ROI, an object with high 121Sb intensity (approx. 3000 CPS; SNR = 560) was found with no detectable 59Co. Furthermore, cavity boundaries showed an increased intensity of 121Sb. This could be caused by leaching Sb from MPs, but we cannot determine if the leaching is caused naturally or by the staining/deparaffinization procedure. This observation needs to be further investigated in future research as a part of toxicology studies. The second ROI (Fig. 3b) revealed high-intensity 121Sb signals, with one fragment exhibiting colocalization of 121Sb and 59Co (approx. 150 CPS; SNR = 2.7). This observation also reveals the same 121Sb intensity inside the boundaries of the tissue. Finally, by registering these elemental overlay maps onto the optical mosaic of the entire tissue section (Fig. 3c), the precise locations of the MPs were confirmed. They were situated within the void spaces of cavities, which are spatially distinct from those of the 127I-rich tissue.
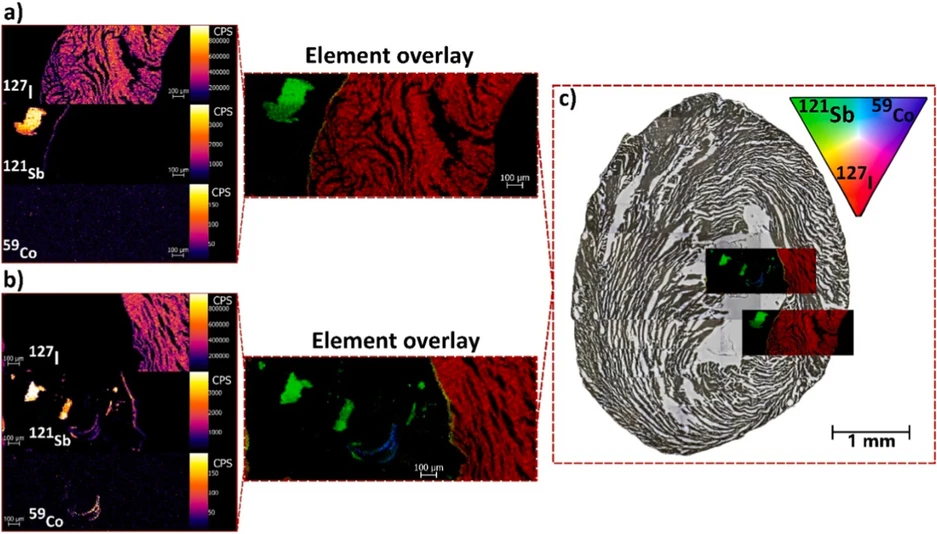 Talanta 308, 2026, 129794: Fig. 3. Identification of chemically distinct Sb- and Co-rich MPs lodged within the tissue cavity using LA-ICP-MS.
Talanta 308, 2026, 129794: Fig. 3. Identification of chemically distinct Sb- and Co-rich MPs lodged within the tissue cavity using LA-ICP-MS.
3.4. Dual modality integration for microplastics imaging in the biological tissue
Integrating μCT with element-specific LA-ICP-MS enabled MP localization within the biological matrix (Fig. 4). Based on density differences relative to the surrounding tissue, the initial plain μCT reconstruction (Fig. 4a) revealed foreign structures inside the tissue cavity. However, as μCT only provides information on density, their structural origin required further verification to confirm MP presence. After referencing the elemental distribution maps (Fig. 4b) with tomographic data, the 121Sb- and 59Co-specific signals confirmed that the foreign objects are indeed MPs. Based on the results of the elemental mapping, MPs were successfully segmented from the surrounding tissue (Fig. 4c), and segmentation was applied to the rest of the volume to generate a comprehensive 3D volumetric model (Fig. 4d). However, it has to be noted that the limitation of this protocol may occur when MPs move from their position during the microtome cutting procedure, especially for the MPs located in the tissue cavity. The MP offset was determined by calculating the average distance from the center of each MP in both datasets, yielding a value of 160 μm without stretching the LA-ICP-MS image. When stretching was applied, which results in possible shape changes during tissue slicing, the mean offset decreased to 123 μm. The offset is visible in our datasets, where the shape of the MPs also does not fully correlate with the μCT slice observation. This step requires precision during tissue handling and transferring from μCT instrumentation into the paraffin medium and subsequent slicing of the tissue, as any organ movement may lead to a slight redistribution of MPs.
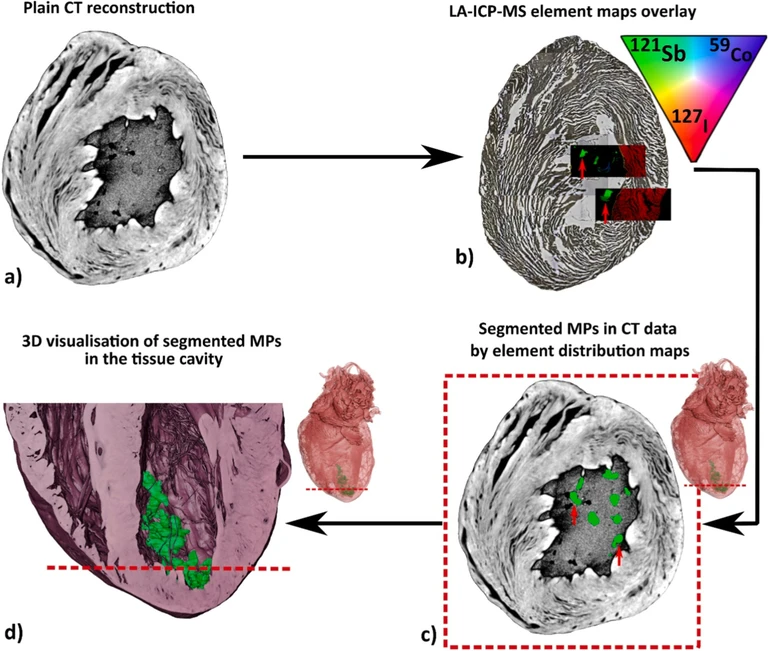 Talanta 308, 2026, 129794: Fig. 4. Dual-modality workflow of using CT and LA-ICP-MS for MP imaging.
Talanta 308, 2026, 129794: Fig. 4. Dual-modality workflow of using CT and LA-ICP-MS for MP imaging.
4. Conclusion
This study successfully demonstrated the efficacy of a correlative imaging workflow combining μCT and LA-ICP-MS for the comprehensive analysis of MPs in soft biological tissue. While μCT provided high-resolution 3D volumetric morphology information, LA-ICP-MS confirmed the chemical composition of MPs by detecting selected metal markers (Sb and Co). For the first time, this combined methodology overcomes the limitations of individual imaging modalities, offering a powerful toolkit for possible 3D assessment and chemical characterization of MPs in biological tissues. However, certain limitations remain, including errors in size distribution due to the potential clustering of MPs within tissue and their dislocation during extensive sample preparation. It is also important to consider that relatively large MPs were used in this study, and the practicality of this approach for smaller MPs, ideally those below 10 μm, which can cross biological membranes and distribute naturally within organisms, still needs to be demonstrated. Nevertheless, this case study shows that the methodology is viable, and future research should focus on its optimization.