Proficiency testing – How much and how often?
Eurachem: Proficiency testing – How much and how often?
A laboratory should have a strategy for its participation in proficiency testing (PT) detailing which schemes it should enrol in (level) and how often (frequency). This is explained in a Eurachem Guide [1].
A balanced selection of tools
Quality related to technical work is dealt with in several ways and is specific to each laboratory. Thus, each laboratory should define its own level and frequency of PT participation after careful analysis of its other quality assurance (QA) measures, such as:
- Regular use of reference materials (RMs) and certified reference materials (CRMs);
- Comparison of results by independent measurement procedures;
- Participation in method development/validation and/or RM characterisation studies;
- Use of internal quality control (IQC) measures;
- Participation in other interlaboratory or intralaboratory comparisons, e.g. analysis of blind samples within the laboratory.
These ‘tools’ are complementary but not perfect and they do not automatically ensure fit-for-purpose results! Important limitations should be identified, e.g. problems in obtaining a stable IQC sample or CRMs/RMs whose composition deviates from that of routine test samples. Also note that legislation may stipulate a minimum frequency of PT participation in certain areas. Frequently, some PT providers offer a flexible participation, e.g. 2, 4, 6 or 12 rounds per year; in rare cases, participation in PT may not be feasible at all.
Areas of technical competence
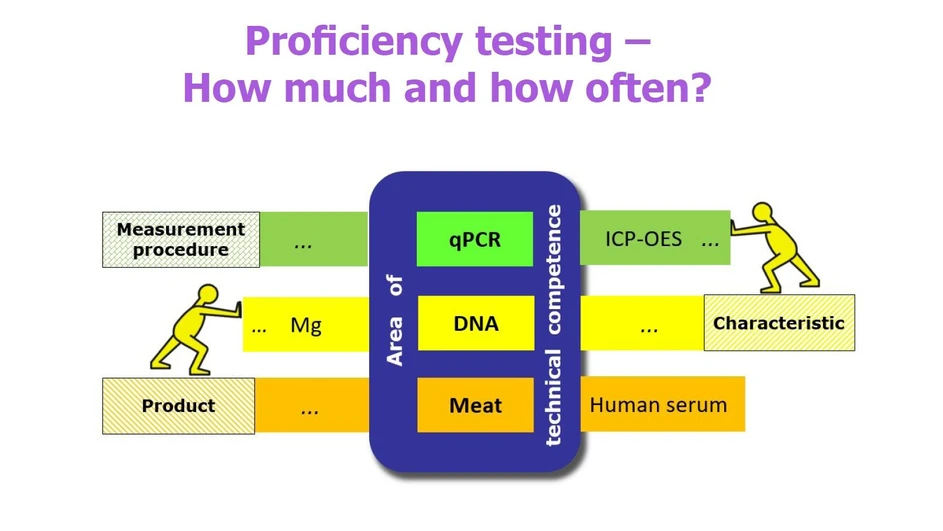
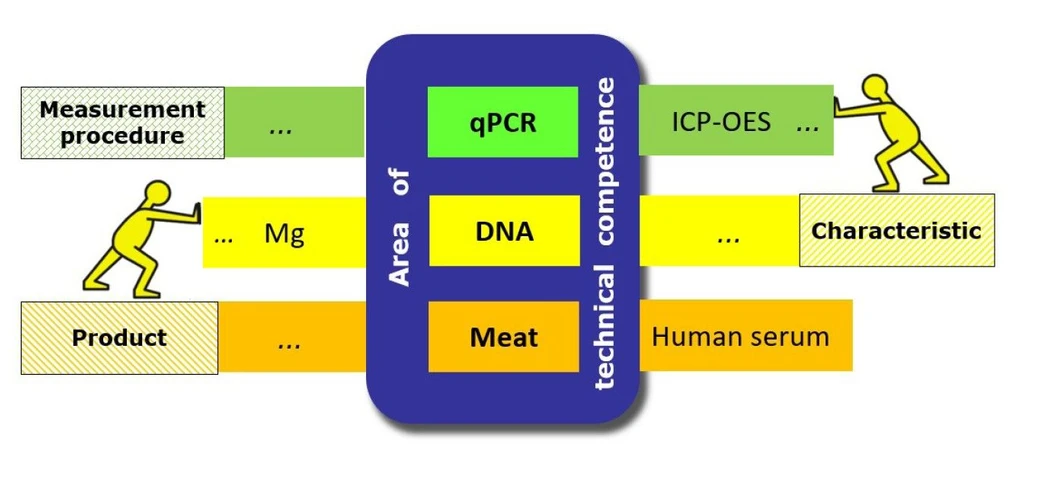
When planning PT participation, the laboratory starts the process by listing its areas of technical competence, defined in terms of three parameters:
- A measurement procedure;
- A characteristic;
- A product.
Two examples are “a measurement procedure using quantitative real-time PCR (qPCR) for the determination of DNA sequences of pathogens in meat” and “a measurement procedure using inductively coupled plasma atomic emission spectroscopy (ICP-AES) for the determination of magnesium concentration in human serum”.
 Eurachem: Areas of technical competence
Eurachem: Areas of technical competence
An area of technical competence may encompass different, but equivalent and comparable, measurement procedures, different characteristics and/or different products. The laboratory can refer to the scope of a standardized procedure, or its method validation data, when planning its level of PT participation. If suitable PT schemes are available, the laboratory is expected to participate at least in a proficiency test related to each of its areas of technical competence.
Risk assessment
In order to decide on a suitable level and frequency of PT participation, the laboratory should conduct a simple risk assessment by considering, for example:
- Limitations in methodology, e.g. instrument instability or interferences from matrix components;
- Extent of validation and/or verification;
- Experience, knowledge and turnover of technical staff;
- Quality and availability of RMs, etc.;
- How the results will be used, e.g. forensic science and environmental control, and the consequences of a wrong result being reported to a customer;
- Number of tests/calibrations/measurements undertaken between PT rounds;
- Complexity of the measurement procedure and changes in requirements, e.g. lower compliance limits.
 Eurachem: Risk assessment
Eurachem: Risk assessment
Case studies
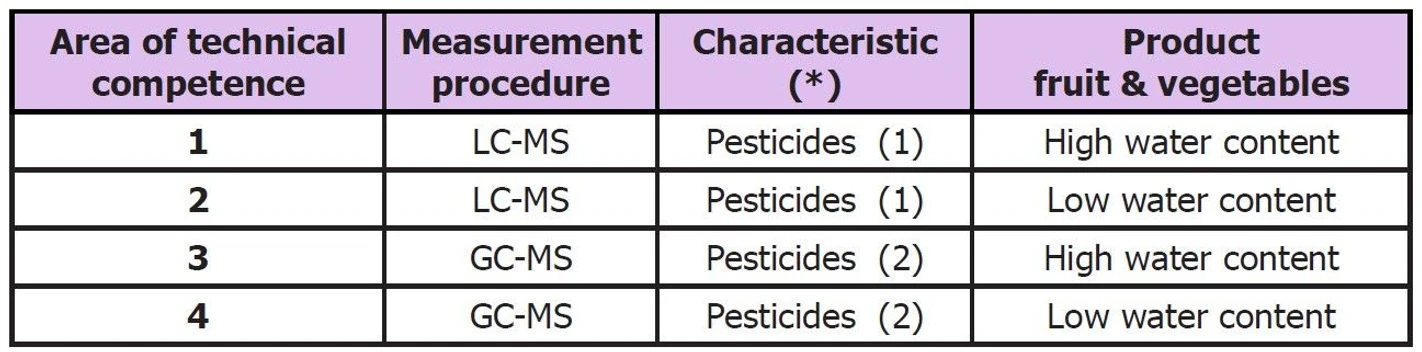
1. A laboratory determines a wide range of pesticides in fruits and vegetables.
Depending on the specific pesticide, the laboratory uses two technically different measurement procedures, LC-MS and/or GC-MS. Additionally, different sample preparation techniques are required depending on whether the matrices are high water content (e.g. cucumbers, pears etc.) or low water content (e.g. chillies, peanuts). Thus, the laboratory would divide its activities into four areas of technical competence for which the laboratory would need to participate in PTs. The laboratory predominately assesses fruits and vegetables of high water content, so it chooses to participate in a PT scheme for high water content fruits/vegetables more frequently.
 Eurachem: Table of Case study 1. * = pesticides analysed in the laboratory by LC-MS (1) or GC-MS (2)
Eurachem: Table of Case study 1. * = pesticides analysed in the laboratory by LC-MS (1) or GC-MS (2)
2. A company has two laboratories, at different sites, both determining a range of minerals and trace elements in various meat, fish and cereal products, using ICP-MS but with different sample preparation techniques for meat/fish matrices and cereal products.
Thus, each laboratory would define its PT participation in terms of two areas of technical competence, both based on an ICP-MS procedure: (i) content of minerals/trace elements in meat/fish; and (ii) content of minerals/ trace elements in cereals. Each laboratory would need to participate in PT schemes that cover both matrices. However, the staff in one laboratory are generally less experienced due to greater staff turnover. The company decides that this laboratory should participate at a greater frequency than the other laboratory.
The PT strategy
Once the level and frequency of participation is established, the PT strategy, as part of the laboratory’s overall quality control plan, is in place. The PT strategy, for an accredited laboratory, could cover, at least, the period between full reassessments, being reviewed on an annual basis. During audits, the laboratory should be prepared to justify the technical arguments that have led to its decision on the “level” and “frequency” of PT participation.
Information about PT providers and schemes can be obtained from your national accreditation body, from the EPTIS website or from other national or international organizations.
[1] B. Brookman and I. Mann (eds.) Eurachem Guide: Selection, Use and Interpretation of Proficiency Testing Schemes (3rd ed. 2021). Available from www.eurachem.org.
EA-4/18 G:2021 - Guidance on the level and frequency of proficiency testing participation. Available from www.european-accreditation.org.